Abstract
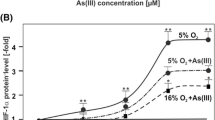
Hypoxia-inducible factors (HIFs), which consist of α and β subunits, are transcription factors involved in regulation of a variety of cellular functions. By blocking the function of the tumor suppressor p53, over-expressions of HIFs are linked to carcinogenesis and tumor progression. Inorganic arsenic, a ubiquitous environmental contaminant, is associated with an increased risk of cancer. Although there are several hypotheses regarding arsenic-induced carcinogenesis, the mechanism of action remains obscure. We have shown that long-term exposure of human bronchial epithelial (HBE) cells to a low level of arsenite increases their proliferation rate and anchorage-independent growth. When introduced into nude mice, the transformed cells are tumorigenic. The present report demonstrates that, with increased time of exposure to arsenite, there is more increased expression of HIF-2α, but not HIF-1α. These factors are known to have different functions, and, in some cases, opposite effects. Arsenite induces accumulation of HIF-2α by inhibiting its degradation through the ubiquitin-mediated proteasome pathway. HIF-2α knockdown, but not HIF-1α knockdown, increases the activation of p53. Finally, inhibition of HIF-2α blocks arsenite-induced proliferation and malignant transformation. Thus, our studies show that blockade of p53 function by inhibiting the ubiquitin-mediated proteasome degradation of HIF-2α, but not that of HIF-1α, is involved in arsenite-induced proliferation and neoplastic transformation of HBE cells.






Similar content being viewed by others
References
Aylon Y, Oren M (2007) Living with p53, dying of p53. Cell 130(4):597–600
Beppu K, Nakamura K, Linehan WM, Rapisarda A, Thiele CJ (2005) Topotecan blocks hypoxia-inducible factor-1 alpha and vascular endothelial growth factor expression induced by insulin-like growth factor-I in neuroblastoma cells. Cancer Res 65(11):4775–4781
Bertout JA, Majmundar AJ, Gordan JD, Lam JC, Ditsworth D, Keith B, Brown EJ, Nathanson KL, Simon MC (2009) HIF2alpha inhibition promotes p53 pathway activity, tumor cell death, and radiation responses. Proc Natl Acad Sci USA 106(34):14391–14396
Chen D, Li M, Luo J, Gu W (2003) Direct interactions between HIF-1 alpha and Mdm2 modulate p53 function. J Biol Chem 278(16):13595–13598
Chen CL, Hsu LI, Chiou HY, Hsueh YM, Chen SY, Wu MM, Chen CJ (2004) Ingested arsenic, cigarette smoking, and lung cancer risk: a follow-up study in arseniasis-endemic areas in Taiwan. JAMA 292(24):2984–2990
Covello KL, Kehler J, Yu H, Gordan JD, Arsham AM, Hu CJ, Labosky PA, Simon MC, Keith B (2006) HIF-2alpha regulates Oct-4: effects of hypoxia on stem cell function, embryonic development, and tumor growth. Genes Dev 20(5):557–570
Cox AD, Der CJ (1994) Biological assays for cellular transformation. Methods Enzymol 238:277–294
Dearth LR, Qian H, Wang T, Baroni TE, Zeng J, Chen SW, Yi SY, Brachmann RK (2007) Inactive full-length p53 mutants lacking dominant wild-type p53 inhibition highlight loss of heterozygosity as an important aspect of p53 status in human cancers. Carcinogenesis 28(2):289–298
Deeble PD, Cox ME, Frierson HF Jr, Sikes RA, Palmer JB, Davidson RJ, Casarez EV, Amorino GP, Parsons SJ (2007) Androgen-independent growth and tumorigenesis of prostate cancer cells are enhanced by the presence of PKA-differentiated neuroendocrine cells. Cancer Res 67(8):3663–3672
Duyndam MC, Hulscher ST, van der Wall E, Pinedo HM, Boven E (2003) Evidence for a role of p38 kinase in hypoxia-inducible factor 1-independent induction of vascular endothelial growth factor expression by sodium arsenite. J Biol Chem 278(9):6885–6895
Florczyk U, Czauderna S, Stachurska A, Tertil M, Nowak W, Kozakowska M, Poellinger L, Jozkowicz A, Loboda A, Dulak J (2010) Opposite effects of HIF-1alpha and HIF-2alpha on the regulation of IL-8 expression in endothelial cells. Free Radic Biol Med 51(10):1882–1892
Franovic A, Lee S (2010) HIF-2alpha: many cancers, one engine? Cell Cycle 9(5):859–860
Gao N, Shen L, Zhang Z, Leonard SS, He H, Zhang XG, Shi X, Jiang BH (2004) Arsenite induces HIF-1alpha and VEGF through PI3 K, Akt and reactive oxygen species in DU145 human prostate carcinoma cells. Mol Cell Biochem 255(1–2):33–45
Gidekel S, Pizov G, Bergman Y, Pikarsky E (2003) Oct-3/4 is a dose-dependent oncogenic fate determinant. Cancer Cell 4(5):361–370
Gordan JD, Bertout JA, Hu CJ, Diehl JA, Simon MC (2007) HIF-2alpha promotes hypoxic cell proliferation by enhancing c-myc transcriptional activity. Cancer Cell 11(4):335–347
Guo W, Yang Z, Xia Q, Liu J, Yu Y, Li J, Zuo Z, Zhang D, Li X, Shi X, Huang C (2010) Arsenite stabilizes HIF-1alpha protein through p85alpha-mediated up-regulation of inducible Hsp70 protein expression. Cell Mol Life Sci 68(3):475–488
Ho E, Ames BN (2002) Low intracellular zinc induces oxidative DNA damage, disrupts p53, NFkappa B, and AP1 DNA binding, and affects DNA repair in a rat glioma cell line. Proc Natl Acad Sci USA 99(26):16770–16775
Hu CJ, Wang LY, Chodosh LA, Keith B, Simon MC (2003) Differential roles of hypoxia-inducible factor 1alpha (HIF-1alpha) and HIF-2alpha in hypoxic gene regulation. Mol Cell Biol 23(24):9361–9374
Hu CJ, Sataur A, Wang L, Chen H, Simon MC (2007) The N-terminal transactivation domain confers target gene specificity of hypoxia-inducible factors HIF-1alpha and HIF-2alpha. Mol Biol Cell 18(11):4528–4542
Huang C, Ke Q, Costa M, Shi X (2004) Molecular mechanisms of arsenic carcinogenesis. Mol Cell Biochem 255(1–2):57–66
Huang HS, Liu ZM, Ding L, Chang WC, Hsu PY, Wang SH, Chi CC, Chuang CH (2006) Opposite effect of ERK1/2 and JNK on p53-independent p21WAF1/CIP1 activation involved in the arsenic trioxide-induced human epidermoid carcinoma A431 cellular cytotoxicity. J Biomed Sci 13(1):113–125
IARC (2004) Some drinking-water disinfectants and contaminants, including arsenic. Monographs on chloramine, chloral and chloral hydrate, dichloroacetic acid, trichloroacetic acid and 3-chloro-4-(dichloromethyl)-5-hydroxy-2(5H)-furanone. IARC Monogr Eval Carcinog Risks Hum 84:269–477
Jing Y, Liu LZ, Jiang Y, Zhu Y, Guo NL, Barnett J, Rojanasakul Y, Agani F, Jiang BH (2011) Cadmium increases HIF-1 and VEGF Expression through ROS, ERK and AKT Signaling Pathways and Induces Malignant Transformation of Human Bronchial Epithelial Cells. Toxicol Sci
Kamat CD, Green DE, Curilla S, Warnke L, Hamilton JW, Sturup S, Clark C, Ihnat MA (2005) Role of HIF signaling on tumorigenesis in response to chronic low-dose arsenic administration. Toxicol Sci 86(2):248–257
Kastan MB (2007) Wild-type p53: tumors can’t stand it. Cell 128(5):837–840
Kuphal S, Winklmeier A, Warnecke C, Bosserhoff AK (2011) Constitutive HIF-1 activity in malignant melanoma. Eur J Cancer 46(6):1159–1169
Li Q, Chen H, Huang X, Costa M (2006) Effects of 12 metal ions on iron regulatory protein 1 (IRP-1) and hypoxia-inducible factor-1 alpha (HIF-1alpha) and HIF-regulated genes. Toxicol Appl Pharmacol 213(3):245–255
Li Y, Xu Y, Ling M, Yang Y, Wang S, Li Z, Zhou J, Wang X, Liu Q (2010) mot-2-Mediated cross talk between nuclear factor-B and p53 is involved in arsenite-induced tumorigenesis of human embryo lung fibroblast cells. Environ Health Perspect 118(7):936–942
Liao WT, Lin P, Cheng TS, Yu HS, Chang LW (2007) Arsenic promotes centrosome abnormalities and cell colony formation in p53 compromised human lung cells. Toxicol Appl Pharmacol 225(2):162–170
Lin Q, Cong X, Yun Z (2010) Differential Hypoxic Regulation of Hypoxia-Inducible Factors 1{alpha} and 2{alpha}. Mol Cancer Res 9(6):757–765
Liu LZ, Jiang Y, Carpenter RL, Jing Y, Peiper SC, Jiang BH (2011) Role and mechanism of arsenic in regulating angiogenesis. PLoS One 6(6):e20858
Lopez-Barneo J, del Toro R, Levitsky KL, Chiara MD, Ortega-Saenz P (2004) Regulation of oxygen sensing by ion channels. J Appl Physiol 96(3):1187–1195; discussion 1170–1182
Nekanti U, Dastidar S, Venugopal P, Totey S, Ta M (2010) Increased proliferation and analysis of differential gene expression in human Wharton’s jelly-derived mesenchymal stromal cells under hypoxia. Int J Biol Sci 6(5):499–512
Ohh M, Park CW, Ivan M, Hoffman MA, Kim TY, Huang LE, Pavletich N, Chau V, Kaelin WG (2000) Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel-Lindau protein. Nat Cell Biol 2(7):423–427
Pi J, Yamauchi H, Kumagai Y, Sun G, Yoshida T, Aikawa H, Hopenhayn-Rich C, Shimojo N (2002) Evidence for induction of oxidative stress caused by chronic exposure of Chinese residents to arsenic contained in drinking water. Environ Health Perspect 110(4):331–336
Reddel RR, Ke Y, Gerwin BI, McMenamin MG, Lechner JF, Su RT, Brash DE, Park JB, Rhim JS, Harris CC (1988) Transformation of human bronchial epithelial cells by infection with SV40 or adenovirus-12 SV40 hybrid virus, or transfection via strontium phosphate coprecipitation with a plasmid containing SV40 early region genes. Cancer Res 48(7):1904–1909
Ryazantseva NV, Novitskii VV, Zhukova OB, Biktasova AK, Chechina OE, Sazonova EV, Radzivil TT, Wice AN, Chasovskikh NY (2010) Role of NF-kB, p53, and p21 in the regulation of TNF-alpha mediated apoptosis of lymphocytes. Bull Exp Biol Med 149(1):50–53
Salnikow K, An WG, Melillo G, Blagosklonny MV, Costa M (1999) Nickel-induced transformation shifts the balance between HIF-1 and p53 transcription factors. Carcinogenesis 20(9):1819–1823
Shen S, Lee J, Weinfeld M, Le XC (2008) Attenuation of DNA damage-induced p53 expression by arsenic: a possible mechanism for arsenic co-carcinogenesis. Mol Carcinog 47(7):508–518
Soucy NV, Klei LR, Mayka DD, Barchowsky A (2004) Signaling pathways for arsenic-stimulated vascular endothelial growth factor-a expression in primary vascular smooth muscle cells. Chem Res Toxicol 17(4):555–563
Treins C, Giorgetti-Peraldi S, Murdaca J, Monthouel-Kartmann MN, Van Obberghen E (2005) Regulation of hypoxia-inducible factor (HIF)-1 activity and expression of HIF hydroxylases in response to insulin-like growth factor I. Mol Endocrinol 19(5):1304–1317
Uchida T, Rossignol F, Matthay MA, Mounier R, Couette S, Clottes E, Clerici C (2004) Prolonged hypoxia differentially regulates hypoxia-inducible factor (HIF)-1alpha and HIF-2alpha expression in lung epithelial cells: implication of natural antisense HIF-1alpha. J Biol Chem 279(15):14871–14878
Warnecke C, Zaborowska Z, Kurreck J, Erdmann VA, Frei U, Wiesener M, Eckardt KU (2004) Differentiating the functional role of hypoxia-inducible factor (HIF)-1alpha and HIF-2alpha (EPAS-1) by the use of RNA interference: erythropoietin is a HIF-2alpha target gene in Hep3B and Kelly cells. Faseb J 18(12):1462–1464
Wen G, Calaf GM, Partridge MA, Echiburu-Chau C, Zhao Y, Huang S, Chai Y, Li B, Hu B, Hei TK (2008) Neoplastic transformation of human small airway epithelial cells induced by arsenic. Mol Med (Cambridge, Mass) 14(1–2):2–10
Wiesener MS, Jurgensen JS, Rosenberger C, Scholze CK, Horstrup JH, Warnecke C, Mandriota S, Bechmann I, Frei UA, Pugh CW, Ratcliffe PJ, Bachmann S, Maxwell PH, Eckardt KU (2003) Widespread hypoxia-inducible expression of HIF-2alpha in distinct cell populations of different organs. Faseb J 17(2):271–273
Xenaki G, Ontikatze T, Rajendran R, Stratford IJ, Dive C, Krstic-Demonacos M, Demonacos C (2008) PCAF is an HIF-1alpha cofactor that regulates p53 transcriptional activity in hypoxia. Oncogene 27(44):5785–5796
Zhang J, Chen GQ (2009) Hypoxia-HIF-1alpha-C/EBPalpha/Runx1 signaling in leukemic cell differentiation. Pathophysiology 16(4):297–303. doi:10.1016/j.pathophys.2009.02.005
Acknowledgments
The authors wish to thank Donald L. Hill (University of Alabama at Birmingham, USA) for editing. This work was supported by the Natural Science Foundations of China (30872146 and 81072327), Research Fund for the Doctoral Program of Higher Education of China (20103234110005), Key Program of Educational Commission of Jiangsu Province of China (11KJA330002), and A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (2010).
Conflict of interest
The authors declare that they have no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yuan Xu and Yuan Li contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, Y., Li, Y., Pang, Y. et al. Blockade of p53 by HIF-2α, but not HIF-1α, is involved in arsenite-induced malignant transformation of human bronchial epithelial cells. Arch Toxicol 86, 947–959 (2012). https://doi.org/10.1007/s00204-012-0810-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-012-0810-x