Abstract
Purpose
Flavanols are an important fraction of our diet both for their antioxidant capacity and because they are constituents of greatly accepted foodstuffs such as tea, wine and cocoa. In addition to their antioxidant activity by directly scavenging intracellular reactive oxygen species (ROS), flavanols have been recently shown to enhance protective enzymes. The objective was to evaluate the antioxidant response of colon-derived Caco2 cells to dietary flavanols.
Methods
Four representative flavanols were selected: epicatechin (EC), epicatechin-3-gallate (ECG), epigallocatechin-3-gallate (EGCG) and procyanidin B2 (PB2). Cell viability, concentration of ROS and reduced glutathione (GSH), and activity of antioxidant/detoxification enzymes and caspase 3 were determined.
Results
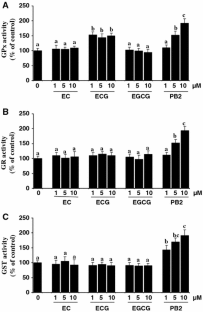
Treatment of Caco2 cells with flavanols decreased ROS production but did not affect GSH content. ECG induced glutathione peroxidase (GPx), whereas PB2 evoked a dose-dependent increase in GPx, glutathione reductase and glutathione-S-transferase. Enhancement of the antioxidant defences implies an improved cell response to an oxidative challenge. Hence, Caco2 cells treated 20 h with the flavanols, especially PB2, and then submitted to an oxidative stress induced by a pro-oxidant, tert-butyl-hydroperoxide, showed a reduced ROS production, restricted activation of caspase 3 and higher viability than cells plainly submitted to the stressor.
Conclusions
Flavanols protect Caco2 cells against an induced oxidative stress and subsequent cellular death by reducing ROS production and preventing caspase-3 activation. In particular, PB2 increases the activity of antioxidant/detoxification enzymes and thus protects Caco2 cells by directly counteracting free radicals and also by activating the antioxidant defence system.




Similar content being viewed by others
References
Sohal RS, Mockett RJ, Orr WC (2002) Mechanisms of aging. An appraisal of the oxidative stress hypothesis. Free Radic Biol Med 33:575–586
Ramos S (2008) Cancer chemoprevention and chemotherapy: dietary polyphenols and signalling pathways. Mol Nutr Food Res 52:507–526
Mukhatar H, Ahmad N (2000) Tea polyphenols: prevention of cancer and optimizing health. Am J Clin Nutr 71:1698S–1702S
Ahn WS, Yoo J, Huh SW, Kim CK, Lee JM, Namkoong SE, Bae SM, Lee IP (2003) Protective effects of green tea extracts (polyphenon E and EGCG) on human cervical lesions. Eur J Cancer Prev 12:383–390
Bettuzzi S, Rizzi F, Belloni L (2007) Clinical relevance of the inhibitory effect of green tea catechins (GtCs) on prostate cancer progression in combination with molecular profiling of catechin-resistant tumors: an integrated view. Pol J Vet Sci 10:57–60
Martín MA, Ramos S, Mateos R, Granado-Serrano AB, Izquierdo-Pulido M, Bravo L, Goya L (2008) Protection of human HepG2 cells against oxidative stress by cocoa phenolic extract. J Agric Food Chem 56:7765–7772
Martín MA, Ramos S, Mateos R, Izquierdo-Pulido M, Bravo L, Goya L (2010) Protection of human HepG2 cells against oxidative stress induced by the flavonoid epicatechin. Phytother Res 24:503–509
Masella R, Di Benedetto R, Varı R, Filesi C, Giovannini C (2005) Novel mechanisms of natural antioxidant compounds in biological systems: involvement of glutathione and glutathione-related enzymes. J Nutr Biochem 16:577–586
Na HK, Surh YJ (2008) Modulation of Nrf2-mediated antioxidant and detoxifying enzyme induction by the green tea polyphenol EGCG. Food Chem Toxicol 46:1271–1278
Alía M, Mateos R, Ramos S, Lecumberri E, Bravo L, Goya L (2006) Influence of quercetin and rutin on growth and the antioxidant defense system in a human hepatoma cell line (HepG2). Eur J Nutr 45:19–28
Molina MF, Sanchez-Reus I, Iglesias I, Benedi J (2003) Quercetin, a flavonoid antioxidant, prevents and protects against ethanol induced oxidative stress in mouse liver. Biol Pharm Bull 26:1398–1402
Martín MA, Granado-Serrano AB, Ramos S, Izquierdo-Pulido M, Bravo L, Goya L (2010) Cocoa flavonoids up-regulate antioxidant enzymes activity via ERK1/2 pathway to protect against oxidative stress-induced apoptosis in HepG2 cells. J Nutr Biochem 21:196–205
Baba S, Osakabe N, Natsume N, Muto Y, Takizawa T, Terao J (2001) In vivo comparison of the bioavailability of catechin, epicatechin and their mixture in orally administered rats. J Nutr 131:2885–2891
Sambuy Y, De Angelis I, Ranaldi G, Scarino ML, Stammati A, Zucco F (2005) The Caco2 cell line as a model of the intestinal barrier: influence of cell and culture-related factors on Caco2 cell functional characteristics. Cell Biol Toxicol 21:1–26
Rufián-Henares JA, Morales FJ (2007) Effect of in vitro enzymatic digestion on antioxidant activity of coffee melanoidins and fractions. J Agric Food Chem 55:10016–10021
Martín MA, Ramos S, Mateos R, Rufián-Henares JA, Morales FJ, Bravo L, Goya L (2009) Biscuit melanoidins of different molecular masses protect human HepG2 cells against oxidative stress. J Agric Food Chem 57:7250–7258
Welder AA, Acosta D (1994) Enzyme leakage as an indicator of cytotoxicity in culture cells. In: Tyson CA, Franzier JM (eds) In vitro toxicity indicators: methods in toxicology. Academic Press, New York, pp 46–49
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27:612–616
Hissin PJ, Hilf R (1976) A fluorometric method for determination of oxidised and reduced glutathione in tissues. Anal Biochem 74:214–226
Gunzler WA, Kramers H, Flohe L (1974) An improved coupled test procedure for glutathione peroxidase. Klin Chem Klin Biochem 12:444
Goldberg DM, Spooner RJ (1987) Glutathione reductase. In: Bergmeyer HV (ed) Methods of enzymatic analysis. Verlag-Chemie, Weinheim, pp 258–265
Herrera B, Fernández M, Alvarez A, Roncero C, Benito M, Gil J, Fabregat I (2001) Activation of caspases occurs downstream from radical oxygen species production, Bcl-xL down-regulation, and early cytochrome C release in apoptosis induced by transforming growth factor beta in rat fetal hepatocytes. Hepatology 34:548–556
Steffen Y, Jung T, Klotz L-O, Schewe T, Grune T, Sies H (2007) Protein modification elicited by oxidized LDL in endothelial cells: protection by epicatechin. Free Radic Biol Med 42:955–970
Natsume M, Osakabe N, Yasuda A, Baba S, Tokunaga T, Kondo K, Osawa T, Terao J (2004) In vitro antioxidative activity of epicatechin glucuronide metabolites present in human and rat plasma. Free Radic Res 38:1341–1348
Jourdain C, Tenca G, Deguercy A, Troplin P, Poelman D (2006) In vitro effects of polyphenols from cocoa and beta sitosterol on the growth of human prostate cancer and normal cells. Eur J Cancer Prev 4:353–361
Ramiro E, Franch A, Castellote C, Pérez-Cano F, Permanyer J, Izquierdo-Pulido M, Castell M (2005) Flavonoids from Theobroma cacao down-regulate inflammatory mediators. J Agric Food Chem 53:8506–8511
Rice-Evans C (2004) Flavonoids and isoflavones: absorption, metabolism and bioactivity. Free Radic Biol Med 36:827–828
Chang W-T, Shao Z-H, Yin J-J, Mehendale S, Wang CZ, Qin Y, Li J, Chen WJ, Chien CT, Becker LB, Van den Hoek TL, Yuan CS (2007) Comparative effects of flavonoids on oxidant scavenging and ischemia-reperfusion injury cardiomyocytes. Eur J Pharmacol 566:58–66
Granado-Serrano AB, Martín MA, Izquierdo-Pulido M, Goya L, Bravo L, Ramos S (2007) Molecular mechanisms of (−)-epicatechin and chlorogenic acid on the regulation of the apoptotic and survival/proliferation pathways in a human hepatoma cell line (HepG2). J Agric Food Chem 55:2020–2027
Arts IC, van de Putte B, Hollman PC (2000) Catechin contents of foods commonly consumed in The Netherlands. 1. Fruits, vegetables, staple foods and processed foods. J Agric Food Chem 48:1746–1751
De Pascual-Teresa S, Santos-Buelga C, Rivas-Gozalo JC (2000) Quantitative analysis of flavan-3-ols in Spanish foodstuffs and beverages. J Agric Food Chem 48:5331–5337
Appeldoorn MM, Vincken JP, Gruppen H, Hollman PC (2009) Procyanidin dimers A1, A2 and B2 are absorbed without conjugation or methylation from the small intestine of rats. J Nutr 139:1469–1473
Luximon-Ramma A, Bahorun T, Crozier A, Zbarsky V, Datla KP, Dexter DT, Aruoma OI (2005) Characterization of the antioxidant functions of flavonoids and proanthocyanidins in Mauritian black teas. Food Res Int 38:357–367
Soobrattee MA, Neergheen VS, Luximon-Ramma A, Aruoma OI, Bahorun T (2005) Phenolics as potential antioxidant therapeutic agents. Mutat Res 579:200–213
Scharf G, Prustomersky S, Knasmuller S, Schulte-Hermann R, Huber WW (2003) Enhancement of glutathione and g-glutamylcysteine synthetase, the rate limiting enzyme of glutathione synthesis, by chemoprotective plant-derived food and beverage components in the human hepatoma cell line HepG2. Nutr Cancer 45:74–83
Lei XG, Cheng W-H, McClung JP (2007) Metabolic regulation and function of glutathione peroxidase-1. Annu Rev Nutr 27:41–61
Argyrou A, Blanchard JS (2004) Flavoprotein disulfide reductases: advances in chemistry and function. Prog Nucleic Acid Res Mol Biol 78:89–142
Masella R, Varı R, D’Archivio M, Di Benedetto R, Mataresse P, Malorni W, Scazzocchio B, Giovannini C (2004) Extra virgin olive oil biophenols inhibit cell-mediated oxidation of LDL by increasing the mRNA transcription of glutathione-related enzymes. J Nutr 134:785–791
Ramiro Puig E, Urpí-Sardá M, Pérez-Cano FJ, Franch A, Castellote C, Andrés-Lacueva C, Izquierdo-Pulido M, Castell M (2008) Cocoa-enriched diet enhances antioxidant enzyme activity and modulates lymphocyte composition in thymus from young rats. J Agric Food Chem 55:6431–6438
Chen C, Kong AN (2004) Dietary cancer-chemopreventive compounds: from signaling and gene expression to pharmacological effects. Free Radic Biol Med 36:1505–1516
Singh R, Czaja MJ (2007) Regulation of hepatocyte apoptosis by oxidative stress. Gastroenterol Hepatol 1:S45–S48
De Ruvo C, Amodio R, Algeri S, Martelli N, Intilangelo A, D′Ancona GM, Esposito E (2000) Nutritional antioxidants as antidegenerative agents. Int J Dev Neurosci 18:359–366
Granado-Serrano AB, Martín MA, Goya L, Bravo L, Ramos S (2009) Time course regulation of survival pathways by epicathechin on HepG2. J Nutr Biochem 20:115–124
Acknowledgments
This work was supported by grant AGL2007-64042/ALI and project CSD 2007-00063 from Programa Consolider-Ingenio from the Spanish Ministry of Education and Science (CICYT). I. Rodríguez-Ramiro is a predoctoral fellow of the JAE predoc program at Consejo Superior de Investigaciones Científicas (CSIC).
Author information
Authors and Affiliations
Corresponding author
Additional information
Both Ildefonso Rodríguez-Ramiro and María Ángeles Martín should be considered as first authors of the article.
Rights and permissions
About this article
Cite this article
Rodríguez-Ramiro, I., Martín, M.Á., Ramos, S. et al. Comparative effects of dietary flavanols on antioxidant defences and their response to oxidant-induced stress on Caco2 cells. Eur J Nutr 50, 313–322 (2011). https://doi.org/10.1007/s00394-010-0139-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-010-0139-2