Abstract
Background
Methylated DNA in fluids may be a suitable biomarker for cancer patients. XAF1 has been shown to be frequently down-regulated in human gastric cancer (GC). Here, we investigated if XAF1 methylation in GC could be a useful biomarker.
Methods
Real-time RT-PCR was used to detect XAF1 mRNA expression; immunohistochemistry and western blot were used to examine XAF1 protein expression in GC tissues (n = 202) and their corresponding para-cancerous histological normal tissues (PCHNTs). Real-time methylation specific-PCR was used to investigate XAF1 promoter methylation in the same panel of GC tissues, their PCHNTs and sera.
Results
We confirmed frequent XAF1 down-regulation in both mRNA and protein levels in GC tissues as compared to normal controls and PCHNTs. XAF1 hypermethylation was evidenced in 83.2% (168/202) of GC tissues and 27.2% (55/202) of PCHNTs, while no methylation was detected in the 88 normal controls. The methylation level in GC tissues was significantly higher than that in PCHNTs (p<0.05). The hypermethylation of XAF1 significantly correlated with the down-regulation of XAF1 in GC tissues in both mRNA and protein levels (p<0.001 each). Moreover, we detected high frequency of XAF1 methylation (69.8%, 141 out of 202) in the sera DNAs from the same patients, while the sera DNAs from 88 non-tumor controls were negative for XAF1 methylation. The XAF1 methylation in both GC tissues and in the sera could be a good biomarker for diagnosis of GC (AUC = 0.85 for tissue and AUC = 0.91 for sera) and significantly correlated with poorer prognosis (p<0.001). In addition, after-surgery negative-to-positive transition of XAF1 methylation in sera strongly associated with tumor recurrence.
Conclusions
1) Dysfunction of XAF1 is frequent and is regulated through XAF1 promoter hypermethylation; 2) Detection of circulating methylated XAF1 DNAs in the serum may be a useful biomarker in diagnosis, evaluating patient’s outcome (prognosis and recurrence) for GC patients.
Introduction
Gastric cancer is one of the most common cancers in China, with a high incidence and mortality, approximately accounting for 10% of all malignancies [1]. Gastric tumorigenesis is a complicated, multiple-step process involving alterations of many genes [2]. Aberrant promoter methylation is one of the major mechanisms to silence some tumor suppressor genes and tumor related genes and plays very important roles in the pathogenesis and progression in human cancers [3], [4]–[6], including gastric cancer [7], [8]. Meanwhile, accumulating data strongly suggested that DNA methylation could be useful and powerful biomarker in cancer risk evaluation [5], [7], early diagnosis [7], predicting patients’ prognosis [7], [8], and evaluating the sensitivity to chemotherapeutic drugs [9]. Our recent study and other researches demonstrated that detecting circulating methylated DNA in blood is a potent and practical approach for cancer patients [7], [10], [11].
X-linked inhibitor of apoptosis (XIAP)-associated factor 1 (XAF1) is a novel negative regulator of XIAP, which reverses XIAP’s protection role on tumor cells [12]–[14]. The loss of XAF1 expression will render tumor cells resistance to apoptosis and promote tumor cell survival [14]–[17]. Dysfunction of XAF1 has been reported in several human cancers probably through promoter methylation [15]–[19], suggesting its importance in tumorigenesis. In human gastric cancer, XAF1 has been reported to be frequently and significantly down-regulated and this down-regulation of XAF1 probably through DNA hypermethylation of specific CpG sites [15], [16], [18]. However, no report is available about XAF1 methylation in blood and its potentiality as a biomarker. In the present study, we examined XAF1 promoter methylation in paired tissue and serum samples from a large panel of patients with gastric cancer, and evaluated circulating methylated XAF1 as a potential biomarker for gastric cancer.
Material and Methods
Ethics Statement
De-identified human tissue samples and sera were obtained from Zhejiang Province Human Tissue Specimen Bank. The use of specimens was approved by the Institutional Review Board at Zhejiang Province Cancer Hospital. Written informed consent was obtained from each patient in accordance with the requirements of our institution’s board of ethics. 88 non-cancer volunteers provided written informed consent. Part of the specimens were from Zhejiang Province People’s Hospital and the First People’s Hospital of Chunan County. The Institutional Review Board on Medical Ethics of Zhejiang Province People’s Hospital and the First People’s Hospital of Chunan County approved the method of specimen collection including written informed consent from all patients, respectively.
Tissue Specimen
Paired tumor and para-cancerous histological normal tissues (PCHNT) specimens were collected at the time of surgery from 202 patients with primary gastric adenocarcinoma at Zhejiang Province Cancer Hospital, Zhejiang Province People’s Hospital and the First People’s Hospital of Chunan County from January 2008 to December 2009. The PCHNT was assessed microscopically for the presence of normal cells and absence of dysplastic cells. None of these cases had undergone any medical treatment before surgery. Demographic, clinical and histopathological parameters of these cases were shown in Table 1. The growth pattern of tumor cells was determined according to Ming’s classification [20]. All recruited patients had been followed-up periodically until the due date. Antral mucosa biopsy specimens from 88 non-cancer volunteers by gastroscopy were randomly collected as controls within the same period, including 54 men and 34 women, with an average age of 52.9 years old. Among these volunteers, 48 patients were diagnosed with chronic non-atrophic gastritis. Meanwhile, paired serum samples were collected before surgery or endoscopy.
Table 1. Clinico-pathological correlations of XAF1 protein expression in gastric cancer tissues.
| Clinicopathological parameters | Number of cases | XAF1 protein expression by IHC | ?2 (p-values) | ||
| High level | Low-moderate level | Absent | |||
| Gender | |||||
| Male | 120 | 23 | 52 | 45 | 1.276 (0.528) |
| Female | 82 | 11 | 40 | 31 | |
| Age at diagnosis | |||||
| <60 | 145 | 23 | 67 | 55 | 0.349 (0.840) |
| ≥60 | 57 | 11 | 25 | 21 | |
| Tumor location | |||||
| Cardia | 63 | 11 | 26 | 26 | 0.712 (0.700) |
| Body/Antrum | 139 | 23 | 66 | 50 | |
| H. pylori infection | |||||
| Negative | 102 | 25 | 47 | 30 | 10.923 (0.04) |
| Positive | 100 | 9 | 45 | 46 | |
| Tumor size | |||||
| <5cm | 92 | 25 | 49 | 18 | 27.588 (p<0.0001) |
| ≥5cm | 110 | 9 | 43 | 58 | |
| Growth pattern | |||||
| Expanding Type | 82 | 12 | 39 | 31 | 0.520 (0.771) |
| Infiltration type | 120 | 22 | 53 | 45 | |
| Histological differentiation | |||||
| Well/moderate | 121 | 29 | 54 | 38 | 12.285 (0.02) |
| Poor | 81 | 5 | 38 | 38 | |
| Lymphatic invasion | |||||
| Negative | 140 | 34 | 76 | 30 | 54.507 (p<0.0001) |
| Positive | 62 | 0 | 16 | 46 | |
| Venous invasion | |||||
| Negative | 148 | 30 | 73 | 45 | 13.293 (0.001) |
| Positive | 54 | 4 | 19 | 31 | |
| Invasive depth | |||||
| T1/T2 | 57 | 21 | 27 | 9 | 29.010 (p<0.0001) |
| T3/T4 | 145 | 13 | 65 | 67 | |
| Lymph node metastasis | |||||
| N0 | 68 | 26 | 40 | 2 | 64.462 (p<0.0001) |
| N1–3 | 134 | 8 | 52 | 74 | |
| Distant metastasis | |||||
| M0 | 184 | 33 | 92 | 59 | 31.575 (p<0.0001) |
| M1 | 18 | 1 | 0 | 17 | |
| TNM stage | |||||
| Stage I/II | 64 | 27 | 37 | 0 | 74.125 (p<0.0001) |
| Stage III/IV | 138 | 7 | 55 | 76 | |
IHC: Immunohistochemistry; ISS: immunohistochemical staining score. High level: ISS 9∼12; Low/Mod level: ISS 1∼8; Absent: ISS 0.
Analysis of Helicobacter Pylori (H. pylori) Infection
Biopsies were obtained from all patients who had endoscopic examination. H. pylori status was determined by rapid Urease test and Giemsa staining methods [21], [22]. It was considered as H. pylori infection when both tests were positive, and the samples with single positive were excluded for statistical analysis [22].
Real-time RT-PCR
The mRNA expression of XAF1 was analyzed by real-time RT-PCR [23]. Total RNAs were extracted using the Trizol (Gibco). A total of 3 µg total RNAs was subjected to reverse transcription using M-MLV reverse transcriptase (Promega). The glyceraldehyde phosphate dehydrogenase (GAPDH) was selected as the internal reference. The sequences of XAF1 primers were as follows: (F) 5′-TGGGTGTAGGATTCTCCAGG-3′, (R) 5′- GGTTTGCCCAAG GACTACAA-3′. GAPDH primer sequences were as follows: (F) 5′-CATGA GAAGTATGACAACAGCCT-3′, (R) 5′-TAATTTTAGGTTAGAGGGTTATTGT- 3′. The 2−ΔΔCt method was used to calculate relative changes in gene expression.
Immunohistochemical Staining
The expression of XAF1 protein was determined by immunohistochemical analysis with XAF1 monoclonal antibody (Santa Cruz Biotechnology). Immunohistochemical staining for XAF1 was carried out using representative paraffin-embedded specimens from the 202 GC patients. After deparaffinization, antigen retrieval in 0.01 M citrate buffer, and inactivation of endogenous peroxidase activity in 3% H2O2/methanol, we incubated the slides with antibody for XAF1 at 4°C overnight, and immunohistochemical staining, following a standard avidinbiotin-peroxidase complex technique, was carried out using 3,3′-diaminobenzidine (DAB) as the chromogen. Nuclei were counterstained with hematoxylin. The immunohistochemical staining score (ISS) is determined by three independent pathologists combining staining frequency and intensity as follows [24], [25]: no staining is scored as 0, 1∼10% of cells stained scored as 1, 11∼50% as 2, 51∼80% as 3, and 81∼100% as 4. Staining intensity is rated on a scale of 0 to 3, with 0, negative; 1, weak; 2, moderate; and 3, strong. When there is multifocal immunoreactivity and there are significant differences in staining intensities between foci, the average of the least intense and most intense staining was recorded. The raw data were converted to the ISS by multiplying the frequency score and the staining intensity score. Theoretically, the ISS could range from 0 to 12. An ISS of 9∼12 was considered strong immunoreactivity, 5∼8 was considered moderate, 1∼4 was considered weak, and 0 was scored as negative. Sections in which the staining could not be well characterized were considered equivocal.
Western Blot Analysis
Paired tumor and PCHNT specimens of 20 cases were randomly selected from 202 gastric cancer patients for western blot analysis. Total protein was extracted and then quantified using the Lowry method [26]. Western blot analysis was performed using anti-XAF1 monoclonal antibodies (Santa Cruz, CA) according to previous report [6]. β-tubulin was served as an internal control.
DNA Extraction, Bisulfite Modification and Real-time Methylation Specific-PCR (MSP)
Serial 5-mm sections that contained carcinoma and non-neoplastic tissues were mounted on non-coated glass slides and dried at 37°C overnight. After deparaffinization and staining with hematoxylin and eosin (H&E), we collected 5,000 nuclei from 5 to 10 serial sections using a 27G needle. The collected nuclei were treated with 40 ml of 200 mg/ml proteinase K (Sigma-Aldrich Co., St. Louis, MO) at 42°C, for 72 hr. The paramagnetic bead technology (AxyPrep Mag Blood gDNA kit, Axygen Scientific, Inc., Union City, CA) was utilized to isolate genomic DNA from fresh blood according to kit’s protocol. The protocol consists of the following step: lysis, binding, washing, and elution. Contaminants are removed during the binding and washing steps. The quality of DNA was assessed by the A260/280 ratio at NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies, Inc., Wilmington, DE), DNA integrity was checked by denaturing agarose gel electrophoresis.
DNAs were modified by sodium bisulfite using the EpiTect Bisulfite kit (Qiagen Inc.) following manufactory’s instructions. Modified DNAs were analyzed by real-time MSP on the ABI7500 PCR (ABI Co.) using the SYBR Premix Taq ExTaq Kit (TaKaRa Co. Ltd). XAF1 methylation and unmethylation specific primers were designed as follows: XAF1 (MF) 5′-TTTGTAAGAAACG AAATTTAATCGA-3′, (MR) 5′- CCTACCCTTAAAACCCACGAT-3′; XAF1(UF) 5′-TTTGTAAGAAATGAAATTTAATTGA-3′, (UR) 5′-CTCCTACCCTTAAAACC CACAAT-3′ [23]. Human genomic DNA (NEB) treated by SssI methyltransferase in vitro was used as a positive control. A peripheral blood DNA from a healthy subject was used as a negative control. The percentage of methylated DNAs in the samples were calculated according to the references as previous described [27], [28]. Methylated DNAs index was scored according to the percentage of DNA methylation; 0, <20%; 1, 20%−40%; 2, 40%−60%; 3, 60%−80%; and 4, >80%. The index score of 0 is considered as DNA unmethylation and scores 1–4 were considered hypermethylated, respectively [7], [27]. The 20% cut off threshold for DNA hypermethylation was based on control normal samples and internal quality controls obtained in the real-time MSP analysis.
Cell Culture and Drugs Treatment
Human gastric cancer cell lines (AGS and KATO-III) were cultured in RPMI1640 medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 U/mL streptomycin at 37°C and 5% CO2, respectively. Cultured cells were treated with 5-aza-2′-deoxycytidine (5-Aza-CdR) (Sigma-Aldrich) at a final concentration of 1.0 µmol/L. Trichostatin A (TSA) (Sigma-Aldrich) at a final concentration of 20 ng/ml was administrated following 5-Aza treatment or alone for 24 h. Cells were collected and subjected to DNA, RNA and protein purification and subsequent analyses.
Statistical Analysis
SPSS 17.0 statistical software was adopted for data analysis. Counting data comparisons between groups were subjected to the χ 2 test and Fisher’s exact test. Survival analysis was computered by means of the Kaplan-Meier method and significant levels were assessed by means of the log-rank test. A univariate analysis with the Cox regression model was used to determine identified prognostic factors, and multivariate analysis with the Cox regression model was used to explore combined effects. For all statistical analyses, p values <0.05 were considered to be statistical significance.
Results
Down-regulation of XAF1 in Primary Gastric Tumors
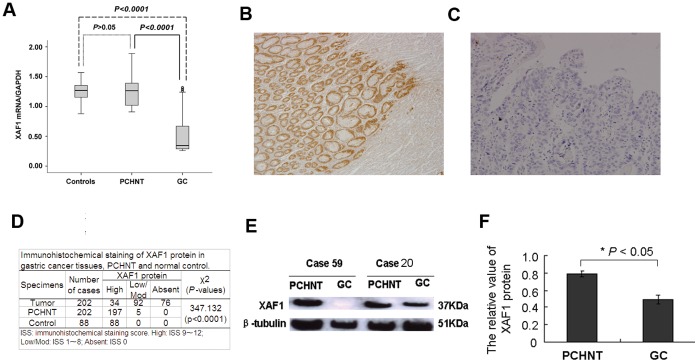
To investigate XAF1 gene expression profile, we examined mRNA expression of XAF1 in 88 non-cancer volunteers, and 202 primary gastric cancer tissues and their corresponding PCHNTs. As shown in Figure 1A, the XAF1 expression was significantly reduced in gastric cancer samples as compared with that in normal controls and PCHNTs (p<0.001). However, there was no significant difference in XAF1 expression in PCHNTs as compared to non-cancer controls.
Figure 1. XAF1 gene expression was down-regulated in gastric cancer (GC) tissues.
A, XAF1 mRNA expression level in GC tissues, PCHNTs (para-cancerous histological normal tissue) and non-cancer controls were determined by real-time RT-PCR and was normalized to GAPDH. B–F, XAF1 protein expression was down-regulated in gastric cancer tissues. B, a representative positive, high expression of XAF1 protein in a PCHNT tissues; C, Absent of XAF1 expression in a poorly differentiated GC; original magnification ×200. D, a summary of XAF1 immunohistochemical staining results in 202 gastric cancers. E, Representative western blot analysis of XAF1 expression in gastric cancer and corresponding PCHNT specimens with different XAF1 methylation levels. PCHNT tissues in Case 59 (XAF1 methylation score: 0); GC tissues in Case 59 (methylated XAF1 score: 4); PCHNT tissues in Case 20 (methylated score: 0); GC tissues in Case 20 (methylated XAF1 score: 1). Beta-tubulin was used as internal control. F, Summary of the western blotting results from 20 GCs and corresponding PCHNTs presented as relative bands density normalized to the beta-tubulin of the same samples.
Furthermore, XAF1 expression was significantly lower in advanced tumors (stage III/IV) than that in early stage tumors (stage I/II) (p<0.0001, Figure S1A), and was significantly lower in poorly differentiated tumors than that in well/moderately differentiated tumors (p<0.0001, Figure S1B).
To check XAF1 protein level in gastric cancer tissues, we performed immunohistochemical analysis in the 202 gastric cancer tissues and their corresponding PCHNTs. Nuclear XAF1 expression was consistently present in the normal gastric epithelia showing high immunoreactive scores. The XAF1 protein expression was detected in high level in 97.5% (197/202) of PCHNTs (Figure 1B). However, high expression of XAF1 protein was only detected in 16.8% (34/202) of gastric cancer tissues; majority of gastric cancer tissues were absent or at a low level of XAF1 protein (Figure 1C). The immunohistochemical findings are summarized in Table 1. The immunohistochemical staining score in gastric cancer tissues was significantly lower than that in PCHNTs or in normal controls (p<0.0001, Figure 1D). The loss of XAF1 protein significantly correlated with H. pylori infection, tumor size, histological differentiation, lymphatic invasion, venous invasion, invasive depth, lymph node metastasis, distant metastasis and clinical stage (Table 1) (all p<0.05).
The immunohistochemical results of 20 gastric cancer tissues were further confirmed by means of western blot analysis. The representative western blotting results in two cases were shown in Figure 1E. The relative quantity of XAF1 protein expression was normalized to the β-tubulin of the same samples. The average XAF1 protein level in 20 gastric cancer tissues was significantly lower than that in PCHNTs (p<0.05) (Figure 1F).
Promoter Hypermethylation of XAF1 in Primary Gastric Tumors
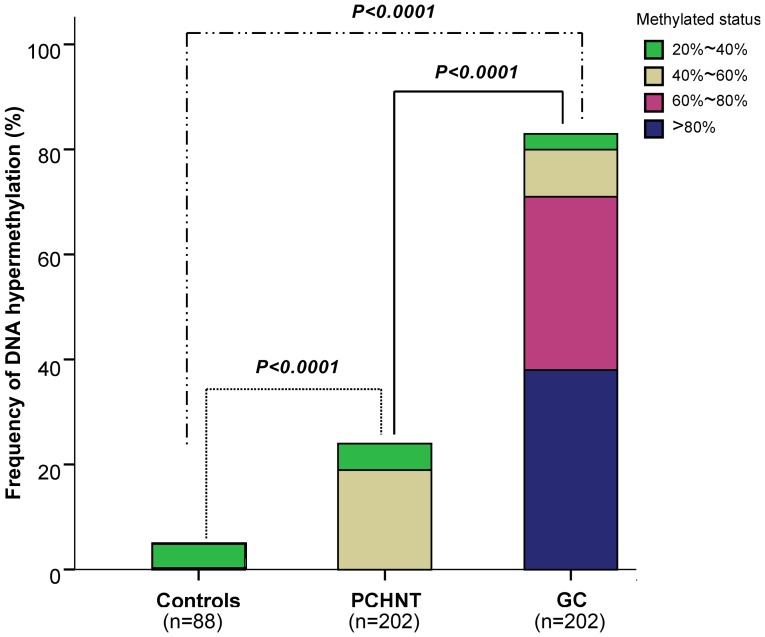
To investigate the molecular mechanisms for the XAF1 silence in gastric cancers, we applied a real-time MSP technology to study the DNA methylation status of XAF1 promoter. The frequency of XAF1 hypermethylation in gastric cancer tissues, corresponding PCHNTs and non-cancer controls were 83.2% (168/202), 24.8% (50/202) and 5.7% (5/88), respectively. The hypermethylation frequency of XAF1 in cancers is significantly higher than that in PCHNTs and non-cancer controls (all p<0.0001, Figure 2).
Figure 2. Summary of XAF1 methylation in 202 gastric cancers tissues, 202 para-cancerous histological normal tissues (PCHNTs) from the same patients and 88 non-cancer volunteers.
Data shows the frequency of XAF1 hypermethylation (DNA methylation level ≥20%) in each group.
Of note, the methylated status of XAF1 significantly correlated with some clinico-pathological parameters in gastric cancer, such as lymph node metastasis, T-stage, H. pylori infection, etc (all p<0.05, Table 2). No significant correlation between the hypermethylation of XAF1 and gender, age at diagnosis, tumor site and distant metastasis was evidenced (all p>0.05) (Table 2).
Table 2. Clinico-pathological correlations of XAF1 promotor hypermethylation in gastric cancer tissues and in sera.
| Clinicopathological parameters | Number of cases | XAF1 in tissues | ?2 (p-values) | XAF1 in serum | ?2 test (p-values) | ||
| M | U | M | U | ||||
| Cases | |||||||
| Tumor | 202 | 168 | 34 | 207.041 (P<0.0001) | 141 | 61 | 283.742 (P<0.0001) |
| PCHNT | 202 | 50 | 152 | ||||
| Normal control | 88 | 5 | 83 | 0 | 88 | ||
| Gender | |||||||
| Male | 120 | 97 | 23 | 1.151 (0.34) | 83 | 37 | 0.057 (0.877) |
| Female | 82 | 71 | 11 | 58 | 24 | ||
| Age at diagnosis | |||||||
| <60 | 145 | 122 | 23 | 0.345 (0.539) | 105 | 40 | 1.663 (0.234) |
| ≥60 | 57 | 46 | 11 | 36 | 21 | ||
| Tumor site | |||||||
| Cardia | 63 | 52 | 11 | 0.026 (0.842) | 43 | 20 | 0.104 (0.744) |
| Body/Antrum | 139 | 116 | 23 | 98 | 41 | ||
| H. pylori | |||||||
| Negative | 102 | 77 | 25 | 8.677 (0.004) | 62 | 40 | 7.949 (0.006) |
| Positive | 100 | 91 | 9 | 79 | 21 | ||
| Tumor size | |||||||
| <5cm | 92 | 67 | 25 | 12.909 (0.001) | 42 | 50 | 46.744 (P<0.0001) |
| ≥5cm | 110 | 101 | 9 | 99 | 11 | ||
| Growth manner | |||||||
| Expanding Type | 82 | 70 | 12 | 0.476 (0.568) | 60 | 22 | 0.743 (0.437) |
| Infiltration type | 120 | 98 | 22 | 81 | 39 | ||
| Histological differentiation | |||||||
| High/Medium | 121 | 92 | 29 | 10.975 (0.001) | 71 | 50 | 17.715 (P<0.0001) |
| Low | 81 | 76 | 5 | 70 | 11 | ||
| Lymphatic invasion | |||||||
| Negative | 140 | 106 | 34 | 18.104 (P<0.0001) | 80 | 60 | 34.678 (P<0.0001) |
| Positive | 62 | 62 | 0 | 61 | 1 | ||
| Venous invasion | |||||||
| Negative | 148 | 118 | 30 | 4.676 (0.034) | 93 | 55 | 12.738 (P<0.0001) |
| Positive | 54 | 50 | 4 | 48 | 6 | ||
| Invasive depth | |||||||
| T1/T2 | 57 | 36 | 21 | 22.714 (P<0.0001) | 16 | 41 | 65.606 (P<0.0001) |
| T3/T4 | 145 | 132 | 13 | 125 | 20 | ||
| Lymph node metastasis | |||||||
| N0 | 68 | 42 | 26 | 33.546 (P<0.0001) | 17 | 51 | 97.612 (P<0.0001) |
| N1–3 | 134 | 126 | 8 | 124 | 10 | ||
| Distant metastasis | |||||||
| M0 | 184 | 151 | 33 | 1.795 (0.319) | 151 | 33 | 1.795 (0.319) |
| M1 | 18 | 17 | 1 | 17 | 1 | ||
| TNM stage | |||||||
| Stage I/II | 64 | 37 | 27 | 43.025 (P<0.0001) | 11 | 53 | 123.032 (P<0.0001) |
| Stage III/IV | 138 | 131 | 7 | 130 | 8 | ||
M: methylation; U: Unmethylation.
XAF1 Promoter Hypermethylation is Associated with its Transcriptional Silencing in Gastric Cancer Cells
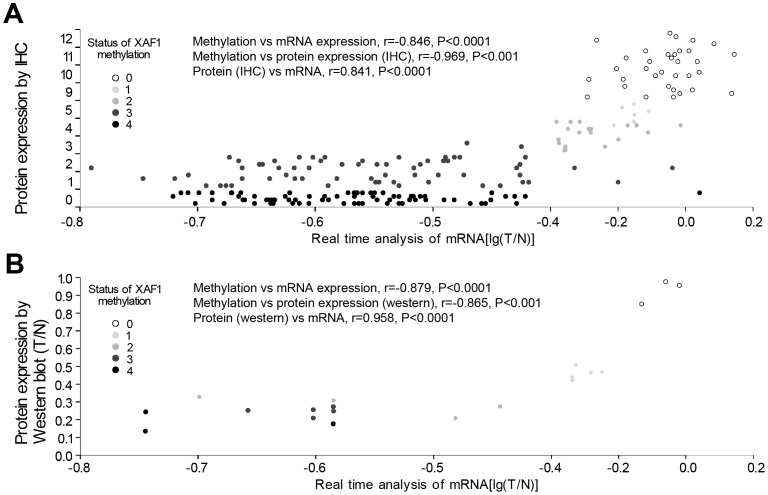
To examine the relationships between XAF1 methylation and XAF1 expression, we compared the XAF1 methylation level with XAF1 mRNA level and protein levels determined either by immunohistochemical analysis (Figure 3A) or western blotting (Figure 3B) by the Spearman correlation analysis. As shown in Figure-3A, XAF1 protein expression (determined by immunohistochemical analysis) in 202 GC tissues was closely correlated with XAF1 mRNA level [lg(T/N)] (determined by RT-PCR) (γ = 0.841, p<0.0001). More important, XAF1 methylation level was significantly associated with XAF1 mRNA level (γ = - 0.846, p<0.0001) and XAF1 protein level (γ = −0.969, p<0.0001). Similar results were obtained when analyzing the correlation of XAF1 promoter methylation and the XAF1 protein expression determined by western blot analysis (Figure 3B). These results strongly indicted that XAF1 expression is regulated by XAF1 promoter methylation.
Figure 3. Correlation of XAF1 methylation with XAF1 expression.
A, Correlation of XAF1 methylation with XAF1 mRNA level determined by RT-PCR analysis and XAF1 protein expression determined by immunohistochemical analysis in 202 gastric cancer tissues. B, Correlation of XAF1 methylation with XAF1 mRNA level determined by RT-PCR analysis and XAF1 protein expression determined by western blotting analysis in 20 frozen gastric cancer tissues. In both A and B, XAF1 methylation scores inversely correlated with XAF1 gene expression in both mRNA and protein levels.
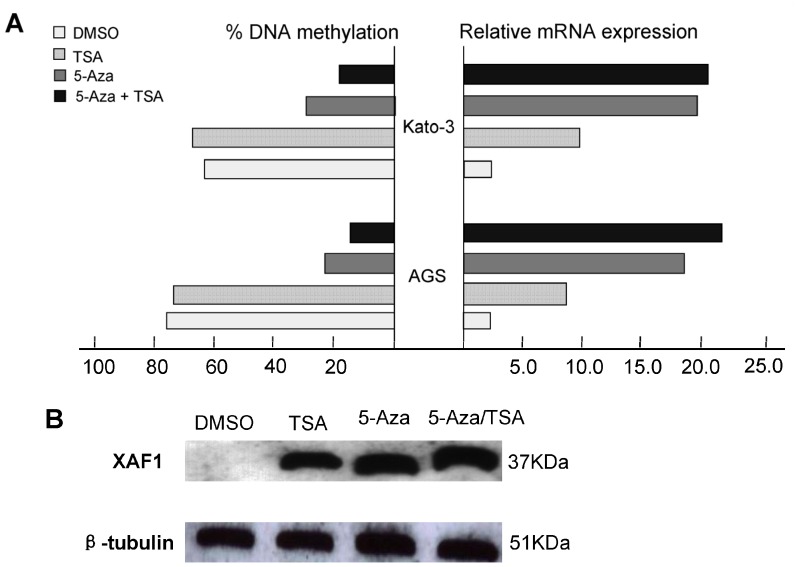
5-Aza-CdR Administration Restores the Expression of XAF1
To further confirm the epigenetic regulation of XAF1 expression, AGS and KATO-III cells were treated with 5-Aza-CdR and/or TSA. XAF1 mRNA expression was reactivated in both gastric cancer cell lines, accompanied by demethylation of XAF1 promoter (Figure 4), indicating that XAF1 is transcriptionally silenced in these cells by DNA hypermethylation. Interestingly, TSA treatment alone was effective in restoring XAF1 expression in AGS and KATO-III without significant change of XAF1 methylation level, suggesting that histone modifications may also be involved in regulating XAF1 expression. However, administration of TSA following 5-Aza-CdR had an additive effect in restoring gene expression with a further decrease in the methylation level of XAF1. These results are in agreement with recent studies that suggested that TSA can have a demethylation effect in a gene-specific manner [5]. The western blot analysis using AGS cells, as a model, confirmed the up-regulation of XAF1 proteins following the 5-Aza-CdR and 5-Aza-CdR/TSA treatments (Figure 4).
Figure 4. A: Transcriptional level of XAF1 gene in gastric cancer cell lines following the 5-Aza-CdR or/and TSA treatments.
Two gastric cancer cell lines (AGS and KATO-III) were treated with 1.0 µmol/L 5-Aza-CdR for 72 hours and/or 100 nM TSA for 24 hours. The methylation levels were determined by real-time MSP. We performed real-time RT-PCR analysis in triplicate for each cDNA sample and used median values in three experiments. The relative XAF1 mRNA expression was normolized to the GAPDH of the same samples using the formula 2−ΔΔCT. The results were multiplied by 100 for a better visualization. The percentage of XAF1 DNA methylation is shown on the left side; whereas the relative mRNA expression of XAF1 is shown on the right side. B: Expression of XAF1 at the protein level following 5-Aza-CdR and TSA treatments. Western blot analysis of AGS cells following treatment with DMSO (control), 5-Aza-CdR, or 5-Aza-CdR/TSA for 72 hours demonstrate up-regulation of the XAF1 proteins in treated cells as compared to control (DMSO). The beta-tubulin is shown as a loading control.
Detection of Circulating XAF1 Methylation
We detected high frequency of XAF1 methylation in primary gastric cancer (83.2%), suggesting that it may be a good biomarker if XAF1 methylation could be detectable in serum. Therefore, we examined XAF1 methylation in the matched serum DNAs from the 202 GC patients. XAF1 methylation was detected in 141 (69.8%) serum DNAs from the 202 gastric cancer patients (Figure 5). In contrast, XAF1 methylation was not detected in serum DNAs from the 88 non-cancer controls.
Figure 5. Compasion of XAF1 methylation between in GC tissues and in corresponding peripheral blood (serum), T: tumor tissues; S: peripheral blood (serum).
Next, we confirmed the consistency in XAF1 methylation between tumor tissues and corresponding serum. In 168 cases that showed XAF1 methylation in tumor tissues, 141 displayed XAF1 methylation in their paired serum, giving a consistency of 83.9% between them. And in the 34 gastric cancer patients without XAF1 methylation in their gastric cancer tissues, no methylation was found in all the serum DNAs (Figure S2).
XAF1 Methylation as a Biomarker in the Diagnosis of Gastric Cancer
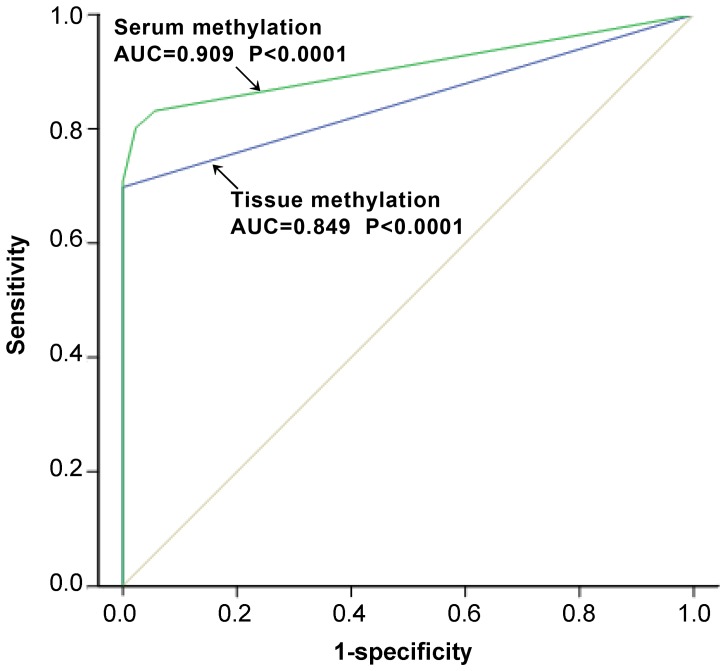
To evaluate the value of XAF1 methylation as a biomarker in diagnosing GC, we plotted receiver operating characteristic (ROC) curves and calculated area under the curve (AUC) values using DNA methylation data from both GC tissues and sera. Both the ROC analyses of XAF1 methylation in tissues and sera revealed significant discriminative capacity (Figure 6); the AUC value of tissues XAF1 methylation was 0.849 (95% confidence interval, 0.806–0.892; p<0.0001), the AUC value of serum AXF1 methylation was 0.909 (95% confidence interval, 0.875–0.942; p<0.0001).
Figure 6. XAF1 methylation as a biomarker for diagnosis of gastric cancer.
Receiver operating characteristic (ROC) curve analysis was used to evaluate the possibility of XAF1 methylation in gastric cancer tissues or in serum as a biomarker to diagnose gastric cancer. For XAF1 methylation in gastric cancer tissues, an area under the ROC curve (AUC) is 0.849 (95% confidence interval, 0.806–0.892; P<0.0001). For XAF1 methylation in serum, the AUC is 0.909 (95% confidence interval, 0.875–0.942; P<0.0001).
In addition, like XAF1 methylation status in gastric cancer tissues and the sera significantly correlated with lymph node metastasis, T-stage, clinical stage, and other clinico-pathological parameters (all p<0.05, Table 2).
XAF1 Methylation Correlated with Prognosis of Gastric Cancer Patients
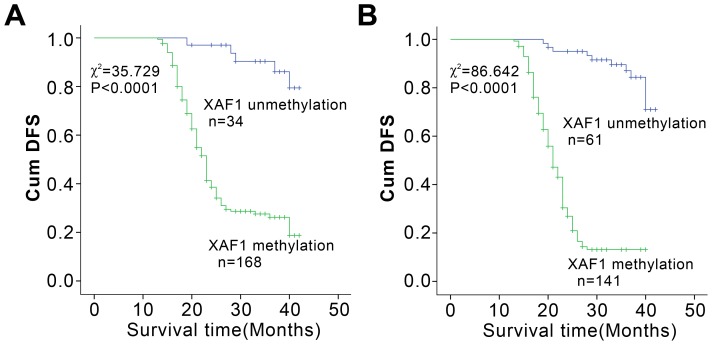
The significant correlation of XAF1 methylation with many clinico-pathological parameters (Table 2) suggested that it may associate with the prognosis of gastric cancer patients. Until the due date of Follow-up, 113 of 168 patients with XAF1 hypermethylation in gastric cancer tissues went rapid disease progression or died. The Median disease free survival (DFS) was only 23.4 months. In contrast, in the 34 patients without XAF1 hypermethylation in their gastric cancer tissues, only 5 patients were deteriorating, and the Median DFS was 39.6 months. Kaplan-Meier analysis demonstrated that patients with XAF1 hypermethylation in their gastric cancer tissues exhibited an obvious worse survival than that without XAF1 hypermethylation (p<0.0001) (Figure 7A). Similarly, Kaplan-Meier analysis proved that patients with positive serum XAF1 methylation had significantly lower DFS than that in the patients without serum XAF1 methylation (p<0.0001) (Figure 7B), indicating that XAF1 promoter methylation in serum was an unfavorable predictor for the gastric cancer patients.
Figure 7. XAF1 methylation in gastric cancer tissues and sera correlated with patients’ prognosis.
Cumulative disease-free survival (Cum DFS) curves are plotted against XAF1 DNA methylation level in gastric cancer tissues (A) and in the sera (B). In both A and B, Kaplan-Meier analysis were used and P<0.0001, respectively.
Cox regression analysis revealed that XAF1 methylation in sera is an independent factor on patients’ survival: patients with XAF1 methylation had worse prognosis (p<0.0001; Hazard ratio, 5.710; 95% CI, 3.474∼9.383). In addition, TNM stages and age at diagnosis could be considered as the influencing factor of prognosis in gastric cancer, only when the effect of XAF1 methylation was eliminated. Moreover, circulating methylated XAF1 in blood had a greater impact on the prognosis than that in TNM stages (Table S1).
Positive Transition of Circulating XAF1 Methylation Predicts Tumor Recurrence and Poor Prognosis
Among the 202 patients who were evaluated for preoperative circulating XAF1 methylation, 72 patients received follow-up examinations of circulating XAF1 methylation 2∼5 times at intervals of 1∼6 months after surgery. Among 12 recurrent patients, 10 patients displayed negative-to-positive transition in XAF1 methylation status in their sera, the other two showed always positive for XAF1 methylation, suggesting that monitoring of XAF1 methylation in serum may be a good marker for predicting tumor recurrence (Table 3).
Table 3. Circulating XAF1 methylation change and patients outcomes.
| The outcome of patients | Circulating XAF1 methylation, after surgery | Total | ||
| Negative always | N to P | Positive always | ||
| DFS | 39 | 0 | 39 | |
| Recurrence | 10 | 2 | 12 | |
| Death | 21 | 21 | ||
| Total | 39 | 10 | 23 | 72 |
DFS: disease-free survival.
N to P: Circulating XAF1 methylation changed from negative to positive in the follow-up after surgery therapy.
Discussion
XAF1 (XIAP-associated factor 1) is a novel XIAP binding protein which could disturb the combining of XIAP with caspases, abolishes its protection on tumor cells and results in tumor cell apoptosis [12]–[14]. The anti-apoptosis function of XIAP is determined by the ratio of expression levels of XIAP against XAF1 [15]. Reduced expression or silence of XAF1 is a frequent event in human tumors [16]–[19]. In the present study, we first confirmed that XAF1 gene expression was significantly down-regulated in both mRNA level and protein level in a large panel of primary gastric cancer tissues, and the down-regulation of XAF1 expression was significantly associated with tumor stages, metastasis and so on, implicating loss of XAF1 function in tumor progression. This is consistent with other research reports [17]–[19]. To explore the molecular mechanisms responsible for the XAF1 silence, we examined XAF1 promoter methylation in a large panel gastric cancer tissues (n = 202) and normal controls (n = 88) using a real-time MSP technology. We detected a high frequency (83.2%) of DNA hypermethylation of XAF1 in gastric cancer tissues. The DNA hypermethylation of XAF1 significant correlated with the down-regulation of XAF1 in both mRNA and protein levels in gastric cancer tissues (Figure 3). In addition, 5-Aza-CdR treatment significantly restored XAF1 expression in XAF1 silenced gastric cancer cell lines (Figure 4). These data strongly suggest that frequent down-regulation of XAF1 in gastric cancer cells is regulated by its promoter hypermethylation. Interestingly, treatment of gastric cancer cells with TSA, a histone deacetylase inhibitor also restored XAF1 expression, alone or combined with 5-Aza-CdR treatment, indicating that histone modification may also be involved in XAF1 regulation.
DNA methylation may be used as a potential tumor biomarker in various human cancers [3]–[11], [23]. The presence of cell-free DNA circulating in peripheral blood has been described in patients with malignant processes, and active release of tumor DNA into the circulation has been reported [29]–[31]. Numerous studies have demonstrated tumor-specific alterations in circulating DNAs recovered from plasma or serum of patients with various malignancies that match genetic changes present in primary tumors, a finding that has potential for molecular diagnosis and prognosis [29]–[32]. The nucleic acid markers described in circulating DNAs include oncogene mutations, microsatellite alterations, gene rearrangements, and epigenetic alterations, such as aberrant promoter hypermethylation [7], [8], [30]–[34]. On the basis of these observations, especially we detected a high frequency of XAF1 DNA methylation in gastric cancer tissues (83.2%), we decided to explore the possibility to use XAF1 promoter methylation in the serum as a biomarker in gastric cancer patients. First we demonstrated a great consistency in detecting XAF1 methylation between sera and primary gastric cancer tissues. Next we analyzed the possibility of using circulating methylated XAF1 as a diagnostic marker using ROC analysis. The AUC (area under curve) of 0.909 demonstrated a high diagnostic value of detecting XAF1 methylation in serum samples. Therefore, our data suggest that detection of XAF1 methylation in circulating DNA could be used as a non-invasive biomarker for diagnosis of gastric cancer. In addition, our results also showed that positive XAF1 methylation in serum was an independent prognostic factor; predicting poor prognosis. More interestingly, transition from negative to positive of circulating XAF1 methylation after surgery was significantly associated with tumor recurrence. These data indicate that detection of XAF1 methylation in circulating serum DNA also can be a tumor biomarker for predicting the gastric cancer patients’ prognosis and for monitoring the tumor recurrence after surgery treatment. Because the number of the patients underwent this follow-up serum examination was small, further research in a large sample size of patients’ number is needed to confirm this interesting finding and to optimize the strategy and protocol for this purpose. In any event, comparing with the traditional ways such as gastroscopy examination for gastric cancer patients, the most remarkable characteristic of circulating methylated XAF1 detection is efficient, rapid, low cost, non-invasive, not destroying environment, high diagnostic conformable rate and so on, and as such demonstrate important application prospects in the future.
Conclusions
Dysfunction of XAF1 is frequent and is regulated through XAF1 promoter hypermethylation in gastric cancer. Circulating methylated XAF1 DNA was associated with tumor burden and malignant progression, which may be a valuable biomarker for diagnosis of gastric cancer, predicting patients’ prognosis and monitoring tumor recurrence after surgery treatment.
Supporting Information
jpg. A, XAF1 gene expression level was lower in advanced gastric cancer (Stage III/IV) than that in early gastric cancer (Stage I/II). B, XAF1 gene expression level was lower in poorly differentiated gastric cancer than that in well/moderately (well/Mod) differentiated gastric cancer, p<0.05.
(TIF)
jpg. Correlation analysis of XAF1 hypermethylation results between GC tissues and paired peripheral blood (sera).
(TIF)
xls. Multivariate survival analysis of clinico-pathological data of 202 gastric carcinoma cases. Cox analysis showed that that XAF1 methylation in sera is an independent factor on patients’ survival (p<0.0001; Hazard ratio, 5.710; 95% CI, 3.474∼9.383). In addition, TNM stages and age at diagnosis could be considered as influencing factor of prognosis for patients with GC.
(DOC)
Acknowledgments
We would like to acknowledge Dr. Ting Zhao, Department of General Surgery, Li XU, Department of Central Laboratory, Zhejiang Province People’s Hospital, and Dr. Ren-Gui Fang, Dr. Jian-Jun Wang, Department of General Surgery, the First People’s Hospital of Chunan County, for collection of the specimens. We also would like to acknowledge Dr. Zhang Gu, Department of Pathology, Zhejiang Province Cancer Hospital, and Dr. Xiao-Hong Yao, Department of Pathology, Xinhua hospital, Medical College, Shanghai Jiaotong University.
Funding Statement
This research was supported by a grant from the Science and Technology General Project of Zhejiang Province (no. 2009C33143), and partly by a grant from the Backbone Talent of Zhejiang Provincial Medicine and Hygiene Platform Programs (no. 2011RCA009). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Zhang H, Sun LL, Meng YL, Song GY, Hu JJ, et al. (2011) Survival trends in gastric cancer patients of Northeast China. World J Gastroenterol 17: 3257–3262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. David S, Meltzer SJ (2010) Stomach-Genetic and epigenetic alterations of preneoplastic and neoplastic lesions. Cancer Biomark 9: 493–507. [DOI] [PubMed] [Google Scholar]
- 3. Sinčić N, Herceg Z (2011) DNA methylation and cancer: ghosts and angels above the genes. Curr Opin Oncol 23: 69–76. [DOI] [PubMed] [Google Scholar]
- 4. Mao WM, Li P, Zheng QQ, Wang CC, Ge MH, et al. (2011) Hypermethylation-modulated downregulation of RASSF1A expression is associated with the progression of esophageal cancer. Arch Med Res 42: 182–188. [DOI] [PubMed] [Google Scholar]
- 5. Peng DF, Razvi M, Chen H, Washington K, Roessner A, et al. (2009) Glutathione peroxidase 7 protects against oxidative DNA damage in oesophageal cells. Gut 58: 5–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ge MH, Chen C, Xu JJ, Ling ZQ (2011) Critical regions and spreading of runt-related transcription factor-3 C-phosphate-G (CpG) island methylation in human salivary gland adenoid cystic carcinoma. Hum Pathol 42: 1862–1872. [DOI] [PubMed] [Google Scholar]
- 7. Lu XX, Yu JL, Ying LS, Han J, Wang S, et al. (2012) Stepwise cumulation of RUNX3 methylation mediated by Helicobacter pylori infection contributes to gastric carcinoma progression. Cancer 118: 5507–5517. [DOI] [PubMed] [Google Scholar]
- 8. Yu QM, Wang XB, Luo J, Wang S, Fang XH, et al. (2012) CDH1 methylation in preoperative peritoneal washes is an independent prognostic factor for gastric cancer. J Surg Oncol 106: 765–771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Charlet J, Schnekenburger M, Brown KW, Diederich M (2012) DNA demethylation increases sensitivity of neuroblastoma cells to chemotherapeutic drugs. Biochem Pharmacol 83: 858–865. [DOI] [PubMed] [Google Scholar]
- 10. Ling ZQ, Zhao Q, Zhou SL, Mao WM (2012) MSH2 promoter hypermethylation in circulating tumor DNA is a valuable predictor of disease-free survival for patients with esophageal squamous cell carcinoma. Eur J Surg Oncol 38: 326–332. [DOI] [PubMed] [Google Scholar]
- 11. Vinayanuwattikun C, Sriuranpong V, Tanasanvimon S, Chantranuwat P, Mutirangura A (2011) Epithelial-specific methylation marker: a potential plasma biomarker in advanced non-small cell lung cancer. J Thorac Oncol 6: 1818–1825. [DOI] [PubMed] [Google Scholar]
- 12. Gyrd-Hansen M, Meier P (2010) IAPs: from caspase inhibitors to modulators of NF-kappaB, inflammation and cancer. Nat Rev Cancer 10: 561–574. [DOI] [PubMed] [Google Scholar]
- 13. Hunter AM, LaCasse EC, Korneluk RG (2007) The inhibitors of apoptosis (IAPs) as cancer targets. Apoptosis 12: 1543–1568. [DOI] [PubMed] [Google Scholar]
- 14. Plenchette S, Cheung HH, Fong WG, LaCasse EC, Korneluk RG (2007) The role of XAF1 in cancer. Curr Opin Investig Drugs 8: 469–476. [PubMed] [Google Scholar]
- 15. Tu SP, Liston P, Cui JT, Lin MC, Jiang XH, et al. (2009) Restoration of XAF1 expression induces apoptosis and inhibits tumor growth in gastric cancer. Int J Cancer 125: 688–697. [DOI] [PubMed] [Google Scholar]
- 16. Zou B, Chim CS, Zeng H, Leung SY, Yang Y, et al. (2006) Correlation between the single-site CpG methylation and expression silencing of the XAF1 gene in human gastric and colon cancers. Gastroenterology 131: 1835–1843. [DOI] [PubMed] [Google Scholar]
- 17. Lee MG, Huh JS, Chung SK, Lee JH, Byun DS, et al. (2006) Promoter CpG hypermethylation and downregulation of XAF1 expression in human urogenital malignancies: implication for attenuated p53 response to apoptotic stresses. Oncogene 25: 5807–5822. [DOI] [PubMed] [Google Scholar]
- 18. Byun DS, Cho K, Ryu BK, Lee MG, Kang MJ, et al. (2003) Hypermethylation of XIAP-associated factor 1, a putative tumor suppressor gene from the 17p13.2 locus, in human gastric adenocarcinomas. Cancer Res 63: 7068–7075. [PubMed] [Google Scholar]
- 19. Kempkensteffen C, Hinz S, Schrader M, Christoph F, Magheli A, et al. (2007) Gene expression and promoter methylation of the XIAP-associated Factor 1 in renal cell carcinomas: correlations with pathology and outcome. Cancer Lett 254: 227–235. [DOI] [PubMed] [Google Scholar]
- 20.Luebke T, Baldus SE, Grass G, Bollschweiler E, Thiele J, et al.. (2005) Histological grading in gastric cancer by Ming classification: correlation with histopathological subtypes, metastasis, and prognosis. World J Surg 9: 1422–1427; discussion 1428. [DOI] [PubMed]
- 21. Hashemi MR, Rahnavardi M, Bikdeli B, Dehghani Zahedani M, Iranmanesh F (2008) Touch cytology in diagnosing Helicobacter pylori: comparison of four staining methods. Cytopathology 19: 179–184. [DOI] [PubMed] [Google Scholar]
- 22. Tian XY, Zhu H, Zhao J, She Q, Zhang GX (2012) Diagnostic performance of urea breath test, rapid urea test, and histology for Helicobacter pylori infection in patients with partial gastrectomy: a meta-analysis. J Clin Gastroenterol 46: 285–292. [DOI] [PubMed] [Google Scholar]
- 23. Liston P, Fong WG, Kelly NL, Toji S, Miyazaki T, et al. (2001) Identification of XAF1 as an antagonist of XIAP anti-Caspase activity. Nat Cell Biol 3: 128–133. [DOI] [PubMed] [Google Scholar]
- 24. Ling ZQ, Li P, Ge MH, Hu FJ, Fang XH, et al. (2011) Aberrant methylation of different DNA repair genes demonstrates distinct prognostic value for esophageal cancer. Dig Dis Sci 56: 2992–3004. [DOI] [PubMed] [Google Scholar]
- 25. Soslow RA, Dannenberg AJ, Rush D, Woerner BM, Khan KN, et al. (2000) COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer 89: 2637–2645. [DOI] [PubMed] [Google Scholar]
- 26. Noble JE, Bailey MJ (2009) Quantitation of protein. Meth Enzymol 463: 73–95. [DOI] [PubMed] [Google Scholar]
- 27. Ling ZQ, Tanaka A, Li P, Nakayama T, Fujiyama Y, et al. (2010) Microsatellite instability with promoter methylation and silencing of hMLH1 can regionally occur during progression of gastric carcinoma. Cancer Lett 297: 244–251. [DOI] [PubMed] [Google Scholar]
- 28. Ling ZQ, Li P, Ge MH, Zhao X, Hu FJ, et al. (2011) Hypermethylation-modulated down-regulation of CDH1 expression contributes to the progression of esophageal cancer. Int J Mol Med 27: 625–635. [DOI] [PubMed] [Google Scholar]
- 29. Yu M (2012) Circulating cell-free mitochondrial DNA as a novel cancer biomarker: opportunities and challenges. Mitochondrial DNA 23: 329–332. [DOI] [PubMed] [Google Scholar]
- 30. Spindler KL, Pallisgaard N, Vogelius I, Jakobsen A (2012) Quantitative cell-free DNA, KRAS, and BRAF mutations in plasma from patients with metastatic colorectal cancer during treatment with cetuximab and irinotecan. Clin Cancer Res 18: 1177–1185. [DOI] [PubMed] [Google Scholar]
- 31. Taniguchi K, Uchida J, Nishino K, Kumagai T, Okuyama T, et al. (2011) Quantitative detection of EGFR mutations in circulating tumor DNA derived from lung adenocarcinomas. Clin Cancer Res 17: 7808–7815. [DOI] [PubMed] [Google Scholar]
- 32. Dawson SJ, Tsui DW, Murtaza M, Biggs H, Rueda OM, et al. (2013) Analysis of circulating tumor DNA to monitor metastatic breast cancer. N Engl J Med 368: 1199–1209. [DOI] [PubMed] [Google Scholar]
- 33. Müller I, Urban K, Pantel K, Schwarzenbach H (2006) Comparison of genetic alterations detected in circulating microsatellite DNA in blood plasma samples of patients with prostate cancer and benign prostatic hyperplasia. Ann N Y Acad Sci 1075: 222–229. [DOI] [PubMed] [Google Scholar]
- 34. He J, Wu J, Jiao Y, Wagner-Johnston N, Ambinder RF, et al. (2011) IgH gene rearrangements as plasma biomarkers in Non- Hodgkin's lymphoma patients. Oncotarget 2: 178–185. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jpg. A, XAF1 gene expression level was lower in advanced gastric cancer (Stage III/IV) than that in early gastric cancer (Stage I/II). B, XAF1 gene expression level was lower in poorly differentiated gastric cancer than that in well/moderately (well/Mod) differentiated gastric cancer, p<0.05.
(TIF)
jpg. Correlation analysis of XAF1 hypermethylation results between GC tissues and paired peripheral blood (sera).
(TIF)
xls. Multivariate survival analysis of clinico-pathological data of 202 gastric carcinoma cases. Cox analysis showed that that XAF1 methylation in sera is an independent factor on patients’ survival (p<0.0001; Hazard ratio, 5.710; 95% CI, 3.474∼9.383). In addition, TNM stages and age at diagnosis could be considered as influencing factor of prognosis for patients with GC.
(DOC)