Attribution Statement: LactMed is a registered trademark of the U.S. Department of Health and Human Services.
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Drugs and Lactation Database (LactMed®) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-.
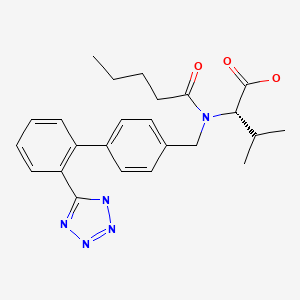
CASRN: 137862-53-4
Drug Levels and Effects
Summary of Use during Lactation
Milk levels after the lowest dose of the combination of valsartan and sacubitril (Entresto) are very low. If the highest recommended maternal dosage (6 times greater) produces proportionate milk levels, they would likely still be quite low. Valsartan is unlikely to affect the nursing infant.
Drug Levels
Maternal Levels. Five mothers taking sacubitril 24 mg and valsartan 26 mg (Entresto) twice daily collected milk samples at 0,1, 2, 4, 6, 8, 10, and 12 hours after taking a dose at steady state. Sacubitril and its active metabolite, LBQ657, were found in the samples, but valsartan was undetectable (<0.19 mcg/L) in all milk samples.[1]
Infant Levels. Relevant published information was not found as of the revision date.
Effects in Breastfed Infants
Two women taking sacubitril 24 mg and valsartan 26 mg (Entresto) did not observe any symptoms in their breastfed infants. Their extent of breastfeeding was not reported.[1]
Effects on Lactation and Breastmilk
Relevant published information was not found as of the revision date.
Alternate Drugs to Consider
References
- 1.
- Falconi S, Okimi A, Wesley S, et al. The concentration of maternal sacubitril/valsartan transferred into human milk is negligible. Front Public Health 2024;12:1389513 [PMC free article: PMC11150817] [PubMed: 38841677]
Substance Identification
Substance Name
Valsartan
CAS Registry Number
137862-53-4
Drug Class
Breast Feeding
Milk, Human
Antihypertensive Agents
Angiotensin II Type 1 Receptor Blockers
Angiotensin Receptor Blockers
ARBs
Disclaimer: Information presented in this database is not meant as a substitute for professional judgment. You should consult your healthcare provider for breastfeeding advice related to your particular situation. The U.S. government does not warrant or assume any liability or responsibility for the accuracy or completeness of the information on this Site.
- User and Medical Advice Disclaimer
- Drugs and Lactation Database (LactMed) - Record Format
- LactMed - Database Creation and Peer Review Process
- Fact Sheet. Drugs and Lactation Database (LactMed)
- Drugs and Lactation Database (LactMed) - Glossary
- LactMed Selected References
- Drugs and Lactation Database (LactMed) - About Dietary Supplements
- Breastfeeding Links
- PubChem SubstanceRelated PubChem Substances
- Review Irbesartan.[Drugs and Lactation Database (...]Review Irbesartan.. Drugs and Lactation Database (LactMed®). 2006
- Review Losartan.[Drugs and Lactation Database (...]Review Losartan.. Drugs and Lactation Database (LactMed®). 2006
- Review Candesartan.[Drugs and Lactation Database (...]Review Candesartan.. Drugs and Lactation Database (LactMed®). 2006
- Review Cilostazol.[Drugs and Lactation Database (...]Review Cilostazol.. Drugs and Lactation Database (LactMed®). 2006
- Review Olmesartan.[Drugs and Lactation Database (...]Review Olmesartan.. Drugs and Lactation Database (LactMed®). 2006
- Valsartan - Drugs and Lactation Database (LactMed®)Valsartan - Drugs and Lactation Database (LactMed®)
- Adenovirus Type 4 and Type 7 Vaccine - Drugs and Lactation Database (LactMed®)Adenovirus Type 4 and Type 7 Vaccine - Drugs and Lactation Database (LactMed®)
- Epoetin Alfa - Drugs and Lactation Database (LactMed®)Epoetin Alfa - Drugs and Lactation Database (LactMed®)
- Succinylcholine - Drugs and Lactation Database (LactMed®)Succinylcholine - Drugs and Lactation Database (LactMed®)
- Solifenacin - Drugs and Lactation Database (LactMed®)Solifenacin - Drugs and Lactation Database (LactMed®)
Your browsing activity is empty.
Activity recording is turned off.
See more...
