Abstract
Purpose
HO-1 has been proved to be associated with tumor aggressivity and poor prognosis in various cancers. Our study provides the first study to demonstrate the relationship of HO-1 expression and clinical characteristics in ovarian cancer patients.
Methods
Immunohistochemistry and western blotting were used to examine the expression of HO-1 in tissue species and fresh tissues. CCK-8 was used to investigate cell viability. Transwell chamber was performed to estimate migration and invasion capacities in A2780 and Skov-3 cells.
Results
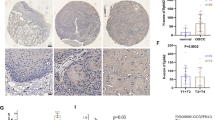
Immunohistochemistry and western blotting showed that the expression of HO-1 was higher in ovarian cancer tissues than normal ovarian tissues. High expression of HO-1 was significantly associated with serous ovarian cancer, high FIGO stage, lymph node metastasis, and non-optimal debulking. Patients with high expression of HO-1 exhibited an unfavorable prognosis. In vitro inducing the expression of HO-1 promoted the proliferation and metastasis of A2780 and Skov-3 cells, with the increased expressions of mesenchymal marker (Vimentin), epithelial–mesenchymal transition-associated transcript factor (Zeb-1), anti-apoptotic protein (Bcl-2), and the decreased expressions of epithelial marker (Keratin) and pro-apoptotic protein (Bax). Meanwhile, after incubating A2780 and Skov-3 together with HO-1 inhibitor, above results could be reversed.
Conclusion
HO-1 might be a potential marker for prediction of ovarian cancer prognosis and a target for ovarian cancer treatment.




Similar content being viewed by others
Change history
20 October 2017
n the original version of the article, Table 2 was incorrect. The corrected Table 2 is shown here. Therefore, in Results (page 3 of original version, right column, line 13), the OR of non-optimal debulking should read OR = 3.036 with 95% CI 1.452–6.348.
References
Siegel R, Miller K, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65(1):5–29.
Maines MD. Heme oxygenase: function, multiplicity, regulatory mechanisms, and clinical applications. FASEB J. 1988;2(10):2557–68.
Cantoni L, Rossi C, Rizzardini M, Gadina M, Ghezzi P. Interleukin-1 and tumour necrosis factor induce hepatic haem oxygenase. Feedback regulation by glucocorticoids. Biochem J. 1991;279(Pt 3):891–4.
Rizzardini M, Terao M, Falciani F, Cantoni L. Cytokine induction of haem oxygenase mRNA in mouse liver. Interleukin 1 transcriptionally activates the haem oxygenase gene. Biochem J. 1993;290(Pt 2):343–7.
Lee PJ, Jiang BH, Chin BY, Iyer NV, Alam J, Semenza GL, et al. Hypoxia-inducible factor-1 mediates transcriptional activation of the heme oxygenase-1 gene in response to hypoxia. J Biol Chem. 1997;272(9):5375–81.
Lee J, Lee SK, Lee BU, Lee HJ, Cho NP, Yoon JH, et al. Upregulation of heme oxygenase-1 in oral epithelial dysplasias. Int J Oral Maxillofac Surg. 2008;37(3):287–92. doi:10.1016/j.ijom.2007.07.028.
Nuhn P, Kunzli BM, Hennig R, Mitkus T, Ramanauskas T, Nobiling R, et al. Heme oxygenase-1 and its metabolites affect pancreatic tumor growth in vivo. Mol Cancer. 2009;8:37. doi:10.1186/1476-4598-8-37.
Kang KA, Maeng YH, Zhang R, Yang YR, Piao MJ, Kim KC, et al. Involvement of heme oxygenase-1 in Korean colon cancer. Tumour Biol J Int Soc Oncodev Biol Med. 2012;33(4):1031–8. doi:10.1007/s13277-012-0336-0.
Gandini NA, Fermento ME, Salomon DG, Obiol DJ, Andres NC, Zenklusen JC, et al. Heme oxygenase-1 expression in human gliomas and its correlation with poor prognosis in patients with astrocytoma. Tumour Biol J Int Soc Oncodev Biol Med. 2014;35(3):2803–15. doi:10.1007/s13277-013-1373-z.
Yin H, Fang J, Liao L, Maeda H, Su Q. Upregulation of heme oxygenase-1 in colorectal cancer patients with increased circulation carbon monoxide levels, potentially affects chemotherapeutic sensitivity. BMC Cancer. 2014;14:436. doi:10.1186/1471-2407-14-436.
Wang TY, Liu CL, Chen MJ, Lee JJ, Pun PC, Cheng SP. Expression of haem oxygenase-1 correlates with tumour aggressiveness and BRAF V600E expression in thyroid cancer. Histopathology. 2015;66(3):447–56. doi:10.1111/his.12562.
Cheng CC, Guan SS, Yang HJ, Chang CC, Luo TY, Chang J, et al. Blocking heme oxygenase-1 by zinc protoporphyrin reduces tumor hypoxia-mediated VEGF release and inhibits tumor angiogenesis as a potential therapeutic agent against colorectal cancer. J Biomed Sci. 2016;23:18. doi:10.1186/s12929-016-0219-6.
Li CG, Pu MF, Li CZ, Gao M, Liu MX, Yu CZ, et al. MicroRNA-1304 suppresses human non-small cell lung cancer cell growth in vitro by targeting heme oxygenase-1. Acta Pharmacol Sin. 2016;. doi:10.1038/aps.2016.92.
Mayerhofer M, Florian S, Krauth MT, Aichberger KJ, Bilban M, Marculescu R, et al. Identification of heme oxygenase-1 as a novel BCR/ABL-dependent survival factor in chronic myeloid leukemia. Can Res. 2004;64(9):3148–54.
Chau LY. Heme oxygenase-1: emerging target of cancer therapy. J Biomed Sci. 2015;22:22. doi:10.1186/s12929-015-0128-0.
Sunamura M, Duda DG, Ghattas MH, Lozonschi L, Motoi F, Yamauchi J, et al. Heme oxygenase-1 accelerates tumor angiogenesis of human pancreatic cancer. Angiogenesis. 2003;6(1):15–24.
Gueron G, De Siervi A, Ferrando M, Salierno M, De Luca P, Elguero B, et al. Critical role of endogenous heme oxygenase 1 as a tuner of the invasive potential of prostate cancer cells. Mol Cancer Res. 2009;7(11):1745–55. doi:10.1158/1541-7786.mcr-08-0325.
Tsai JR, Wang HM, Liu PL, Chen YH, Yang MC, Chou SH, et al. High expression of heme oxygenase-1 is associated with tumor invasiveness and poor clinical outcome in non-small cell lung cancer patients. Cell Oncol (Dordr). 2012;35(6):461–71. doi:10.1007/s13402-012-0105-5.
Wegiel B, Gallo D, Csizmadia E, Harris C, Belcher J, Vercellotti GM, et al. Carbon monoxide expedites metabolic exhaustion to inhibit tumor growth. Can Res. 2013;73(23):7009–21. doi:10.1158/0008-5472.can-13-1075.
Degese MS, Mendizabal JE, Gandini NA, Gutkind JS, Molinolo A, Hewitt SM, et al. Expression of heme oxygenase-1 in non-small cell lung cancer (NSCLC) and its correlation with clinical data. Lung Cancer (Amst Neth). 2012;77(1):168–75. doi:10.1016/j.lungcan.2012.02.016.
Lee SS, Yang SF, Tsai CH, Chou MC, Chou MY, Chang YC. Upregulation of heme oxygenase-1 expression in areca-quid-chewing-associated oral squamous cell carcinoma. J Formosan Med Assoc Taiwan yi zhi. 2008;107(5):355–63. doi:10.1016/s0929-6646(08)60100-x.
Becker JC, Fukui H, Imai Y, Sekikawa A, Kimura T, Yamagishi H, et al. Colonic expression of heme oxygenase-1 is associated with a better long-term survival in patients with colorectal cancer. Scand J Gastroenterol. 2007;42(7):852–8. doi:10.1080/00365520701192383.
Yanagawa T, Omura K, Harada H, Nakaso K, Iwasa S, Koyama Y, et al. Heme oxygenase-1 expression predicts cervical lymph node metastasis of tongue squamous cell carcinomas. Oral Oncol. 2004;40(1):21–7.
Joung EJ, Li MH, Lee HG, Somparn N, Jung YS, Na HK, et al. Capsaicin induces heme oxygenase-1 expression in HepG2 cells via activation of PI3K-Nrf2 signaling: NAD(P)H:quinone oxidoreductase as a potential target. Antioxid Redox Signal. 2007;9(12):2087–98. doi:10.1089/ars.2007.1827.
Boschetto P, Zeni E, Mazzetti L, Miotto D, Lo Cascio N, Maestrelli P, et al. Decreased heme-oxygenase (HO)-1 in the macrophages of non-small cell lung cancer. Lung Cancer (Amst Neth). 2008;59(2):192–7. doi:10.1016/j.lungcan.2007.08.019.
Lin PH, Lan WM, Chau LY. TRC8 suppresses tumorigenesis through targeting heme oxygenase-1 for ubiquitination and degradation. Oncogene. 2013;32(18):2325–34. doi:10.1038/onc.2012.244.
Chen GG, Liu ZM, Vlantis AC, Tse GM, Leung BC, van Hasselt CA. Heme oxygenase-1 protects against apoptosis induced by tumor necrosis factor-alpha and cycloheximide in papillary thyroid carcinoma cells. J Cell Biochem. 2004;92(6):1246–56. doi:10.1002/jcb.20157.
Liu YS, Li HS, Qi DF, Zhang J, Jiang XC, Shi K, et al. Zinc protoporphyrin IX enhances chemotherapeutic response of hepatoma cells to cisplatin. World J Gastroenterol WJG. 2014;20(26):8572–82. doi:10.3748/wjg.v20.i26.8572.
Kocanova S, Buytaert E, Matroule JY, Piette J, Golab J, de Witte P, et al. Induction of heme-oxygenase 1 requires the p38MAPK and PI3K pathways and suppresses apoptotic cell death following hypericin-mediated photodynamic therapy. Apoptosis Int J Programm Cell Death. 2007;12(4):731–41. doi:10.1007/s10495-006-0016-x.
Cannito S, Novo E, di Bonzo LV, Busletta C, Colombatto S, Parola M. Epithelial-mesenchymal transition: from molecular mechanisms, redox regulation to implications in human health and disease. Antioxid Redox Signal. 2010;12(12):1383–430. doi:10.1089/ars.2009.2737.
Acknowledgements
This work was supported by grants from the Development and Reform commission Project of Shandong Province (26010104081103).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Ethical approval
For this type of study, formal consent is not required.
Additional information
A correction to this article is available online at https://doi.org/10.1007/s12094-017-1769-0.
Rights and permissions
About this article
Cite this article
Zhao, Z., Xu, Y., Lu, J. et al. High expression of HO-1 predicts poor prognosis of ovarian cancer patients and promotes proliferation and aggressiveness of ovarian cancer cells. Clin Transl Oncol 20, 491–499 (2018). https://doi.org/10.1007/s12094-017-1738-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-017-1738-7