Journal Description
Bioengineering
Bioengineering
is an international, scientific, peer-reviewed, open access journal on the science and technology of bioengineering, published monthly online by MDPI. The Society for Regenerative Medicine (Russian Federation) (RPO) is affiliated with Bioengineering and its members receive discounts on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, CAPlus / SciFinder, Inspec, and other databases.
- Journal Rank: JCR - Q2 (Engineering, Biomedical)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17.7 days after submission; acceptance to publication is undertaken in 3.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
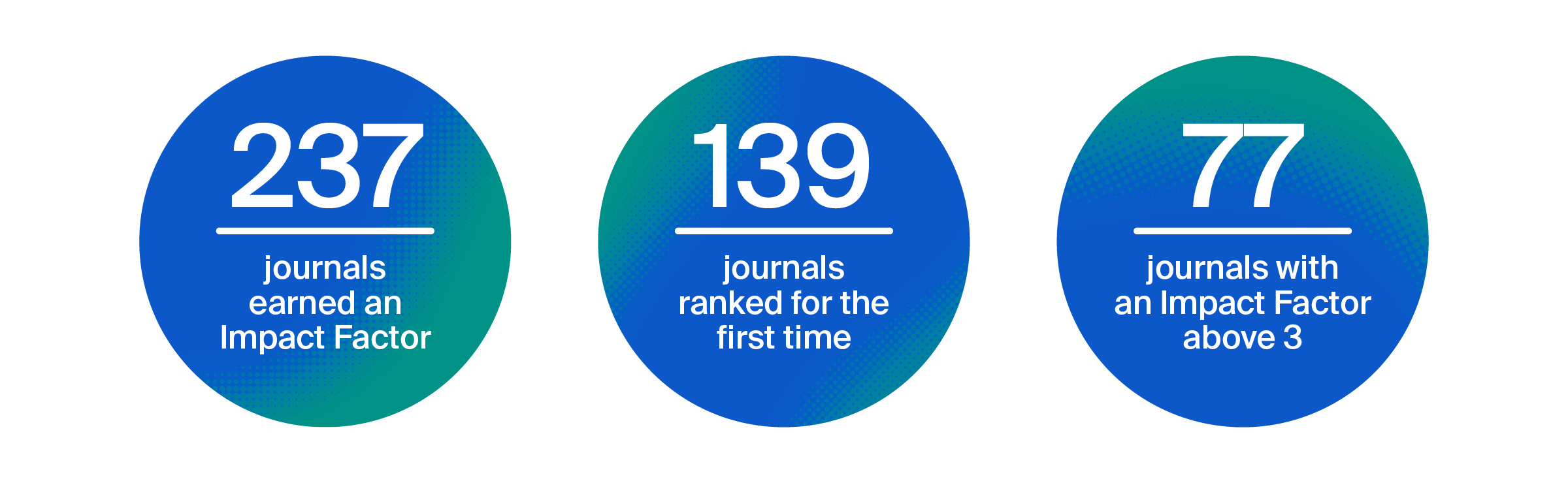
Impact Factor:
3.8 (2023)
Latest Articles
Alveolar Ridge Preservation Using a Novel Species-Specific Collagen-Enriched Deproteinized Bovine Bone Mineral: Histological Evaluation of a Prospective Case Series
Bioengineering 2024, 11(7), 665; https://doi.org/10.3390/bioengineering11070665 (registering DOI) - 28 Jun 2024
Abstract
In recent years, the significance of maintaining the alveolar ridge following tooth extractions has markedly increased. Alveolar ridge preservation (ARP) is a commonly utilized technique and a variety of bone substitute materials and biologics are applied in different combinations. For this purpose, a
[...] Read more.
In recent years, the significance of maintaining the alveolar ridge following tooth extractions has markedly increased. Alveolar ridge preservation (ARP) is a commonly utilized technique and a variety of bone substitute materials and biologics are applied in different combinations. For this purpose, a histological evaluation and the clinical necessity of subsequent guided bone regeneration (GBR) in delayed implantations were investigated in a prospective case series after ARP with a novel deproteinized bovine bone material (95%) in combination with a species-specific collagen (5%) (C-DBBM). Notably, block-form bone substitutes without porcine collagen are limited, and moreover, the availability of histological data on this material remains limited. Ten patients, each scheduled for tooth extraction and desiring future implantation, were included in this study. Following tooth extraction, ARP was performed using a block form of C-DBBM in conjunction with a double-folded bovine cross-linked collagen membrane (xCM). This membrane was openly exposed to the oral cavity and secured using a crisscross suture. After a healing period ranging from 130 to 319 days, guided trephine drilling was performed for implant insertion utilizing static computer-aided implant surgery (s-CAIS). Cores harvested from the area previously treated with ARP were histologically processed and examined. Guided bone regeneration (GBR) was not necessary for any of the implantations. Histological examination revealed the development of a lattice of cancellous bone trabeculae through appositional membranous osteogenesis at various stages surrounding C-DBBM granules as well as larger spongy or compact ossicles with minimal remnants. The clinical follow-up period ranged from 2.5 to 4.5 years, during which no biological or technical complications occurred. Within the limitations of this prospective case series, it can be concluded that ARP using this novel C-DBBM in combination with a bovine xCM could be a treatment option to avoid the need for subsequent GBR in delayed implantations with the opportunity of a bovine species-specific biomaterial chain.
Full article
(This article belongs to the Section Regenerative Engineering)
Open AccessCommentary
Bridging the Gap: Integrating 3D Bioprinting and Microfluidics for Advanced Multi-Organ Models in Biomedical Research
by
Marco De Spirito, Valentina Palmieri, Giordano Perini and Massimiliano Papi
Bioengineering 2024, 11(7), 664; https://doi.org/10.3390/bioengineering11070664 (registering DOI) - 28 Jun 2024
Abstract
Recent advancements in 3D bioprinting and microfluidic lab-on-chip systems offer promising solutions to the limitations of traditional animal models in biomedical research. Three-dimensional bioprinting enables the creation of complex, patient-specific tissue models that mimic human physiology more accurately than animal models. These 3D
[...] Read more.
Recent advancements in 3D bioprinting and microfluidic lab-on-chip systems offer promising solutions to the limitations of traditional animal models in biomedical research. Three-dimensional bioprinting enables the creation of complex, patient-specific tissue models that mimic human physiology more accurately than animal models. These 3D bioprinted tissues, when integrated with microfluidic systems, can replicate the dynamic environment of the human body, allowing for the development of multi-organ models. This integration facilitates more precise drug screening and personalized therapy development by simulating interactions between different organ systems. Such innovations not only improve predictive accuracy but also address ethical concerns associated with animal testing, aligning with the three Rs principle. Future directions include enhancing bioprinting resolution, developing advanced bioinks, and incorporating AI for optimized system design. These technologies hold the potential to revolutionize drug development, regenerative medicine, and disease modeling, leading to more effective, personalized, and humane treatments.
Full article
(This article belongs to the Section Biomedical Engineering and Biomaterials)
Open AccessArticle
Pretreatment Sarcopenia and MRI-Based Radiomics to Predict the Response of Neoadjuvant Chemotherapy in Triple-Negative Breast Cancer
by
Jiamin Guo, Wenjun Meng, Qian Li, Yichen Zheng, Hongkun Yin, Ying Liu, Shuang Zhao and Ji Ma
Bioengineering 2024, 11(7), 663; https://doi.org/10.3390/bioengineering11070663 (registering DOI) - 28 Jun 2024
Abstract
The association between sarcopenia and the effectiveness of neoadjuvant chemotherapy (NAC) in triple-negative breast cancer (TNBC) remains uncertain. This study aims to examine the potential of sarcopenia as a predictive factor for the response to NAC in TNBC, and to assess whether its
[...] Read more.
The association between sarcopenia and the effectiveness of neoadjuvant chemotherapy (NAC) in triple-negative breast cancer (TNBC) remains uncertain. This study aims to examine the potential of sarcopenia as a predictive factor for the response to NAC in TNBC, and to assess whether its combination with MRI radiomic signatures can improve the predictive accuracy. We collected clinical and pathological information, as well as pretreatment breast MRI and abdominal CT images, of 121 patients with TNBC who underwent NAC at our hospital between January 2012 and September 2021. The presence of pretreatment sarcopenia was assessed using the L3 skeletal muscle index. Clinical models were constructed based on independent risk factors identified by univariate regression analysis. Radiomics data were extracted on breast MRI images and the radiomics prediction models were constructed. We integrated independent risk factors and radiomic features to build the combined models. The results of this study demonstrated that sarcopenia is an independent predictive factor for NAC efficacy in TNBC. The combination of sarcopenia and MRI radiomic signatures can further improve predictive performance.
Full article
(This article belongs to the Special Issue Computational Biology and Biostatistics for Public Health)
Open AccessArticle
Identification of Calculous Pyonephrosis by CT-Based Radiomics and Deep Learning
by
Guanjie Yuan, Lingli Cai, Weinuo Qu, Ziling Zhou, Ping Liang, Jun Chen, Chuou Xu, Jiaqiao Zhang, Shaogang Wang, Qian Chu and Zhen Li
Bioengineering 2024, 11(7), 662; https://doi.org/10.3390/bioengineering11070662 (registering DOI) - 28 Jun 2024
Abstract
Urgent detection of calculous pyonephrosis is crucial for surgical planning and preventing severe outcomes. This study aims to evaluate the performance of computed tomography (CT)-based radiomics and a three-dimensional convolutional neural network (3D-CNN) model, integrated with independent clinical factors, to identify patients with
[...] Read more.
Urgent detection of calculous pyonephrosis is crucial for surgical planning and preventing severe outcomes. This study aims to evaluate the performance of computed tomography (CT)-based radiomics and a three-dimensional convolutional neural network (3D-CNN) model, integrated with independent clinical factors, to identify patients with calculous pyonephrosis. We recruited 182 patients receiving either percutaneous nephrostomy tube placement or percutaneous nephrolithotomy for calculous hydronephrosis or pyonephrosis. The regions of interest were manually delineated on plain CT images and the CT attenuation value (HU) was measured. Radiomics analysis was performed using least absolute shrinkage and selection operator (LASSO). A 3D-CNN model was also developed. The better-performing machine-learning model was combined with independent clinical factors to build a comprehensive clinical machine-learning model. The performance of these models was assessed using receiver operating characteristic analysis and decision curve analysis. Fever, blood neutrophils, and urine leukocytes were independent risk factors for pyonephrosis. The radiomics model showed higher area under the curve (AUC) than the 3D-CNN model and HU (0.876 vs. 0.599, 0.578; p = 0.003, 0.002) in the testing cohort. The clinical machine-learning model surpassed the clinical model in both the training (0.975 vs. 0.904, p = 0.019) and testing (0.967 vs. 0.889, p = 0.045) cohorts.
Full article
(This article belongs to the Section Biomedical Engineering and Biomaterials)
►▼
Show Figures

Graphical abstract

Figure 2

Figure 3

Figure 4

Figure 5

Figure 6
Open AccessArticle
Extracellular Overexpression of a Neutral Pullulanase in Bacillus subtilis through Multiple Copy Genome Integration and Atypical Secretion Pathway Enhancement
by
Wenkang Dong, Xiaoping Fu, Dasen Zhou, Jia Teng, Jun Yang, Jie Zhen, Xingya Zhao, Yihan Liu, Hongchen Zheng and Wenqin Bai
Bioengineering 2024, 11(7), 661; https://doi.org/10.3390/bioengineering11070661 (registering DOI) - 28 Jun 2024
Abstract
Neutral pullulanases, having a good application prospect in trehalose production, showed a limited expression level. In order to address this issue, two approaches were utilized to enhance the yield of a new neutral pullulanase variant (PulA3E) in B. subtilis. One involved using
[...] Read more.
Neutral pullulanases, having a good application prospect in trehalose production, showed a limited expression level. In order to address this issue, two approaches were utilized to enhance the yield of a new neutral pullulanase variant (PulA3E) in B. subtilis. One involved using multiple copies of genome integration to increase its expression level and fermentation stability. The other focused on enhancing the PulA-type atypical secretion pathway to further improve the secretory expression of PulA3E. Several strains with different numbers of genome integrations, ranging from one to four copies, were constructed. The four-copy genome integration strain PD showed the highest extracellular pullulanase activity. Additionally, the integration sites ytxE, ytrF, and trpP were selected based on their ability to enhance the PulA-type atypical secretion pathway. Furthermore, overexpressing the predicated regulatory genes comEA and yvbW of the PulA-type atypical secretion pathway in PD further improved its extracellular expression. Three-liter fermenter scale-up production of PD and PD-ARY yielded extracellular pullulanase activity of 1767.1 U/mL at 54 h and 2465.1 U/mL at 78 h, respectively. Finally, supplementing PulA3E with 40 U/g maltodextrin in the multi-enzyme catalyzed system resulted in the highest trehalose production of 166 g/L and the substrate conversion rate of 83%, indicating its potential for industrial application.
Full article
(This article belongs to the Section Biochemical Engineering)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Integrating PointNet-Based Model and Machine Learning Algorithms for Classification of Rupture Status of IAs
by
Yilu Shou, Zhenpeng Chen, Pujie Feng, Yanan Wei, Beier Qi, Ruijuan Dong, Hongyu Yu and Haiyun Li
Bioengineering 2024, 11(7), 660; https://doi.org/10.3390/bioengineering11070660 (registering DOI) - 28 Jun 2024
Abstract
Background: The rupture of intracranial aneurysms (IAs) would result in subarachnoid hemorrhage with high mortality and disability. Predicting the risk of IAs rupture remains a challenge. Methods: This paper proposed an effective method for classifying IAs rupture status by integrating a PointNet-based model
[...] Read more.
Background: The rupture of intracranial aneurysms (IAs) would result in subarachnoid hemorrhage with high mortality and disability. Predicting the risk of IAs rupture remains a challenge. Methods: This paper proposed an effective method for classifying IAs rupture status by integrating a PointNet-based model and machine learning algorithms. First, medical image segmentation and reconstruction algorithms were applied to 3D Digital Subtraction Angiography (DSA) imaging data to construct three-dimensional IAs geometric models. Geometrical parameters of IAs were then acquired using Geomagic, followed by the computation of hemodynamic clouds and hemodynamic parameters using Computational Fluid Dynamics (CFD). A PointNet-based model was developed to extract different dimensional hemodynamic cloud features. Finally, five types of machine learning algorithms were applied on geometrical parameters, hemodynamic parameters, and hemodynamic cloud features to classify and recognize IAs rupture status. The classification performance of different dimensional hemodynamic cloud features was also compared. Results: The 16-, 32-, 64-, and 1024-dimensional hemodynamic cloud features were extracted with the PointNet-based model, respectively, and the four types of cloud features in combination with the geometrical parameters and hemodynamic parameters were respectively applied to classify the rupture status of IAs. The best classification outcomes were achieved in the case of 16-dimensional hemodynamic cloud features, the accuracy of XGBoost, CatBoost, SVM, LightGBM, and LR algorithms was 0.887, 0.857, 0.854, 0.857, and 0.908, respectively, and the AUCs were 0.917, 0.934, 0.946, 0.920, and 0.944. In contrast, when only utilizing geometrical parameters and hemodynamic parameters, the accuracies were 0.836, 0.816, 0.826, 0.832, and 0.885, respectively, with AUC values of 0.908, 0.922, 0.930, 0.884, and 0.921. Conclusion: In this paper, classification models for IAs rupture status were constructed by integrating a PointNet-based model and machine learning algorithms. Experiments demonstrated that hemodynamic cloud features had a certain contribution weight to the classification of IAs rupture status. When 16-dimensional hemodynamic cloud features were added to the morphological and hemodynamic features, the models achieved the highest classification accuracies and AUCs. Our models and algorithms would provide valuable insights for the clinical diagnosis and treatment of IAs.
Full article
(This article belongs to the Section Biomedical Engineering and Biomaterials)
►▼
Show Figures

Graphical abstract

Figure 2

Figure 3

Figure 4

Figure 5

Figure 6
Open AccessArticle
Validation of Pelvis and Trunk Range of Motion as Assessed Using Inertial Measurement Units
by
Farwa Ali, Cecilia A. Hogen, Emily J. Miller and Kenton R. Kaufman
Bioengineering 2024, 11(7), 659; https://doi.org/10.3390/bioengineering11070659 - 28 Jun 2024
Abstract
Trunk and pelvis range of motion (ROM) is essential to perform activities of daily living. The ROM may become limited with aging or with neuromusculoskeletal disorders. Inertial measurement units (IMU) with out-of-the box software solutions are increasingly being used to assess motion. We
[...] Read more.
Trunk and pelvis range of motion (ROM) is essential to perform activities of daily living. The ROM may become limited with aging or with neuromusculoskeletal disorders. Inertial measurement units (IMU) with out-of-the box software solutions are increasingly being used to assess motion. We hypothesize that the accuracy (validity) and reliability (consistency) of the trunk and pelvis ROM during steady-state gait in normal individuals as measured using the Opal APDM 6 sensor IMU system and calculated using Mobility Lab version 4 software will be comparable to a gold-standard optoelectric motion capture system. Thirteen healthy young adults participated in the study. Trunk ROM, measured using the IMU was within 5–7 degrees of the motion capture system for all three planes and within 10 degrees for pelvis ROM. We also used a triad of markers mounted on the sternum and sacrum IMU for a head-to-head comparison of trunk and pelvis ROM. The IMU measurements were within 5–10 degrees of the triad. A greater variability of ROM measurements was seen for the pelvis in the transverse plane. IMUs and their custom software provide a valid and reliable measurement for trunk and pelvis ROM in normal individuals, and important considerations for future applications are discussed.
Full article
(This article belongs to the Special Issue Biomechanics of Human Movement and Its Clinical Applications)
►▼
Show Figures

Figure 1

Figure 3

Figure 4

Figure 5
Open AccessArticle
The Approach to Sensing the True Fetal Heart Rate for CTG Monitoring: An Evaluation of Effectiveness of Deep Learning with Doppler Ultrasound Signals
by
Yuta Hirono, Ikumi Sato, Chiharu Kai, Akifumi Yoshida, Naoki Kodama, Fumikage Uchida and Satoshi Kasai
Bioengineering 2024, 11(7), 658; https://doi.org/10.3390/bioengineering11070658 - 28 Jun 2024
Abstract
Cardiotocography (CTG) is widely used to assess fetal well-being. CTG is typically obtained using ultrasound and autocorrelation methods, which extract periodicity from the signal to calculate the heart rate. However, during labor, maternal vessel pulsations can be measured, resulting in the output of
[...] Read more.
Cardiotocography (CTG) is widely used to assess fetal well-being. CTG is typically obtained using ultrasound and autocorrelation methods, which extract periodicity from the signal to calculate the heart rate. However, during labor, maternal vessel pulsations can be measured, resulting in the output of the maternal heart rate (MHR). Since the autocorrelation output is displayed as fetal heart rate (FHR), there is a risk that obstetricians may mistakenly evaluate the fetal condition based on MHR, potentially overlooking the necessity for medical intervention. This study proposes a method that utilizes Doppler ultrasound (DUS) signals and artificial intelligence (AI) to determine whether the heart rate obtained by autocorrelation is of fetal origin. We developed a system to simultaneously record DUS signals and CTG and obtained data from 425 cases. The midwife annotated the DUS signals by auditory differentiation, providing data for AI, which included 30,160 data points from the fetal heart and 2160 data points from the maternal vessel. Comparing the classification accuracy of the AI model and a simple mathematical method, the AI model achieved the best performance, with an area under the curve (AUC) of 0.98. Integrating this system into fetal monitoring could provide a new indicator for evaluating CTG quality.
Full article
(This article belongs to the Section Biosignal Processing)
►▼
Show Figures

Figure 1

Figure 3

Figure 4

Figure 5
Open AccessArticle
Identifying First-Trimester Risk Factors for SGA-LGA Using Weighted Inheritance Voting Ensemble Learning
by
Sau Nguyen Van, Jinhui Cui, Yanling Wang, Hui Jiang, Feng Sha and Ye Li
Bioengineering 2024, 11(7), 657; https://doi.org/10.3390/bioengineering11070657 - 27 Jun 2024
Abstract
The classification of fetuses as Small for Gestational Age (SGA) and Large for Gestational Age (LGA) is a critical aspect of neonatal health assessment. SGA and LGA, terms used to describe fetal weights that fall below or above the expected weights for Appropriate
[...] Read more.
The classification of fetuses as Small for Gestational Age (SGA) and Large for Gestational Age (LGA) is a critical aspect of neonatal health assessment. SGA and LGA, terms used to describe fetal weights that fall below or above the expected weights for Appropriate for Gestational Age (AGA) fetuses, indicate intrauterine growth restriction and excessive fetal growth, respectively. Early prediction and assessment of latent risk factors associated with these classifications can facilitate timely medical interventions, thereby optimizing the health outcomes for both the infant and the mother. This study aims to leverage first-trimester data to achieve these objectives. This study analyzed data from 7943 pregnant women, including 424 SGA, 928 LGA, and 6591 AGA cases, collected from 2015 to 2021 at the Third Affiliated Hospital of Sun Yat-sen University in Guangzhou, China. We propose a novel algorithm, named the Weighted Inheritance Voting Ensemble Learning Algorithm (WIVELA), to predict the classification of fetuses into SGA, LGA, and AGA categories based on biochemical parameters, maternal factors, and morbidity during pregnancy. Additionally, we proposed algorithms for relevance determination based on the classifier to ascertain the importance of features associated with SGA and LGA. The proposed classification solution demonstrated a notable average accuracy rate of
(This article belongs to the Section Biosignal Processing)
►▼
Show Figures

Figure 1

Figure 3

Figure 4

Figure 5

Figure 6

Figure 7

Figure 8

Figure 9
Open AccessSystematic Review
Mesenchymal Stromal Cells for the Enhancement of Surgical Flexor Tendon Repair in Animal Models: A Systematic Review and Meta-Analysis
by
Ilias Ektor Epanomeritakis, Andreas Eleftheriou, Anna Economou, Victor Lu and Wasim Khan
Bioengineering 2024, 11(7), 656; https://doi.org/10.3390/bioengineering11070656 - 27 Jun 2024
Abstract
►▼
Show Figures
Flexor tendon lacerations are primarily treated by surgical repair. Limited intrinsic healing ability means the repair site can remain weak. Furthermore, adhesion formation may reduce range of motion post-operatively. Mesenchymal stromal cells (MSCs) have been trialled for repair and regeneration of multiple musculoskeletal
[...] Read more.
Flexor tendon lacerations are primarily treated by surgical repair. Limited intrinsic healing ability means the repair site can remain weak. Furthermore, adhesion formation may reduce range of motion post-operatively. Mesenchymal stromal cells (MSCs) have been trialled for repair and regeneration of multiple musculoskeletal structures. Our goal was to determine the efficacy of MSCs in enhancing the biomechanical properties of surgically repaired flexor tendons. A PRISMA systematic review was conducted using four databases (PubMed, Ovid, Web of Science, and CINAHL) to identify studies using MSCs to augment surgical repair of flexor tendon injuries in animals compared to surgical repair alone. Nine studies were included, which investigated either bone marrow- or adipose-derived MSCs. Results of biomechanical testing were extracted and meta-analyses were performed regarding the maximum load, friction and properties relating to viscoelastic behaviour. There was no significant difference in maximum load at final follow-up. However, friction, a surrogate measure of adhesions, was significantly reduced following the application of MSCs (p = 0.04). Other properties showed variable results and dissipation of the therapeutic benefits of MSCs over time. In conclusion, MSCs reduce adhesion formation following tendon injury. This may result from their immunomodulatory function, dampening the inflammatory response. However, this may come at the cost of favourable healing which will restore the tendon’s viscoelastic properties. The short duration of some improvements may reflect MSCs’ limited survival or poor retention. Further investigation is needed to clarify the effect of MSC therapy and optimise its duration of action.
Full article

Figure 1

Figure 3

Figure 4

Figure 5

Figure 6

Figure 7

Figure 8