Journal Description
Pharmacoepidemiology
Pharmacoepidemiology
is an international, peer-reviewed, open access journal on high-quality epidemiological, clinical research across the fields of clinical pharmacology and epidemiology, published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 34.2 days after submission; acceptance to publication is undertaken in 4.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Pharmacoepidemiology is a companion journal of Pharmaceuticals.
Latest Articles
A Retrospective Analysis of the Clinical Effectiveness of Tigecycline in the Treatment of Clostridioides difficile-Associated Diarrhea
Pharmacoepidemiology 2024, 3(2), 231-240; https://doi.org/10.3390/pharma3020015 - 13 Jun 2024
Abstract
Clostridioides difficile infection (CDI) is the leading cause of nosocomial diarrhea in the United States. Tigecycline has been proposed as a potential treatment for CDI, though limited clinical data exist to support this practice. The objective of this study was to determine if
[...] Read more.
Clostridioides difficile infection (CDI) is the leading cause of nosocomial diarrhea in the United States. Tigecycline has been proposed as a potential treatment for CDI, though limited clinical data exist to support this practice. The objective of this study was to determine if the provision of tigecycline provides a clinically meaningful benefit to inpatients with CDI. This study was a retrospective chart review enrolling inpatients receiving treatment for CDI. Patients were divided into cohorts depending on whether they received a standard antibiotic therapy regimen for CDI or an antibiotic treatment regimen that included tigecycline. The primary outcome was clinical recovery at the time of hospital discharge. A total of 39 and 22 patients were included in the standard antibiotic therapy and tigecycline groups, respectively. ATLAS (Age, Treatment, Leukocyte, Albumin, Serum creatinine) scores at the time of CDI diagnosis were similar between the two groups, though patients in the tigecycline groups were more likely to represent a recurrent episode of CDI. There was no difference in the rate of clinical recovery at the time of hospital discharge between the standard antibiotic therapy and tigecycline groups (38.5% vs. 36.4%, p = 0.8710). These data do not support the routine use of tigecycline for the treatment of CDI, though interpretation is limited due to baseline differences between groups and the retrospective, observational nature of this study.
Full article
(This article belongs to the Special Issue Anti-Infectives: Pharmacoepidemiology and Clinical Pharmacology)
►
Show Figures
Open AccessCommunication
Ocular Chloramphenicol Exposure in Early Childhood in Aotearoa/New Zealand
by
Isabella M. Y. Cheung, Simon Horsburgh, Ewan Smith, Samantha Simkin and Akilesh Gokul
Pharmacoepidemiology 2024, 3(2), 223-230; https://doi.org/10.3390/pharma3020014 - 14 May 2024
Abstract
►▼
Show Figures
Background: The paediatric use of ophthalmic chloramphenicol in New Zealand (NZ) is relatively high; however, little more is known about its utilisation, including whether this is equitable. This study aimed to describe chloramphenicol utilisation in NZ children aged five years and under, by
[...] Read more.
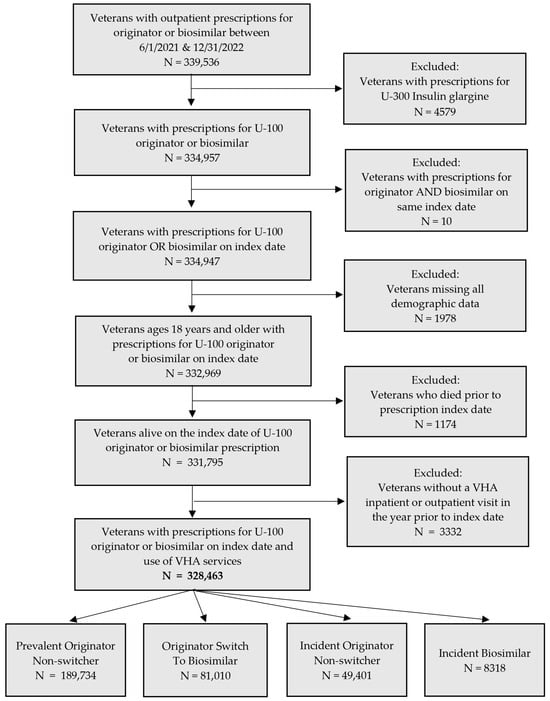
Background: The paediatric use of ophthalmic chloramphenicol in New Zealand (NZ) is relatively high; however, little more is known about its utilisation, including whether this is equitable. This study aimed to describe chloramphenicol utilisation in NZ children aged five years and under, by patient ethnicity, socioeconomic deprivation, and urban/non-urban domicile. Methods: This analysis included every publicly subsidised chloramphenicol dispensing received from birth to five years of age, for every child born in NZ in 2013. Cumulative proportion of first exposure, dispensing rate per person-year, and seasonality of dispensing were quantified. These were calculated following stratification by ethnicity, socioeconomic deprivation quintile, and urban/non-urban health district. For cumulative proportion of first exposure, odds ratios (OR) were calculated and multivariate logistic regression was performed. For dispensing rate, incidence rate ratios (IRR) were calculated and zero-inflated Poisson regression was performed. Results: Almost one-quarter of NZ children received their first dispensing within the first year of life. By five years of age, 55.2% of children had received their first dispensing. By five years of age, children of Pacific ethnicity, those in the highest deprivation quintile, and in those non-urban health districts had lower odds of receiving chloramphenicol (adjusted OR 0.90, 0.79, and 0.81, respectively, all p < 0.001). In contrast, children of Māori ethnicity had higher odds (adjusted OR 1.99, p < 0.001). Māori and Pacific ethnicity, and residence in non-urban health districts, were associated with fewer dispensings (adjusted IRR 0.88, 0.75 and 0.87, all p < 0.001). In contrast, deprivation quintile was not significantly associated with dispensing rate. Conclusion: Chloramphenicol utilisation is prevalent among NZ children, and utilisation may be lower among children of Pacific ethnicity and those in non-urban areas
Full article

Figure 1
Open AccessReview
Adverse Drug Reactions in Multimorbid Older People Exposed to Polypharmacy: Epidemiology and Prevention
by
Siobhán McGettigan, Denis Curtin and Denis O’Mahony
Pharmacoepidemiology 2024, 3(2), 208-222; https://doi.org/10.3390/pharma3020013 - 30 Apr 2024
Abstract
Adverse drug reactions (ADRs) are frequent and represent a significant healthcare burden. ADRs are a potentially avoidable contributor to excess unscheduled hospital admissions, higher morbidity, mortality, and healthcare costs. The objective of this review is to examine the epidemiology of ADRs in older
[...] Read more.
Adverse drug reactions (ADRs) are frequent and represent a significant healthcare burden. ADRs are a potentially avoidable contributor to excess unscheduled hospital admissions, higher morbidity, mortality, and healthcare costs. The objective of this review is to examine the epidemiology of ADRs in older multimorbid adults and to explore strategies for ADR prevention. ADRs in this population are often linked to commonly prescribed medications, including anticoagulants, antiplatelet agents, insulin, and non-steroidal anti-inflammatory drugs, but ADRs and adverse drug events (ADEs) in fact encompass a much broader range of culprit drugs. Age-related factors such as changes in pharmacokinetics and pharmacodynamics, multimorbidity, polypharmacy, and frailty have been associated with ADR occurrences. Various strategies have been proposed to prevent ADRs in different clinical settings, such as structured routine medication review and the use of bespoke software applications to identify potentially inappropriate prescriptions and drug interactions. Although these approaches have demonstrated some improvement in the quality of prescribing, there is still a lack of consistent evidence regarding their effectiveness in preventing ADRs. The nuanced and often intricate complexities associated with older patients’ pharmacotherapy necessitate a comprehensive approach to attenuate the impact of ADRs within this growing section of most populations globally.
Full article
(This article belongs to the Special Issue Feature Papers of Pharmacoepidemiology)
►▼
Show Figures
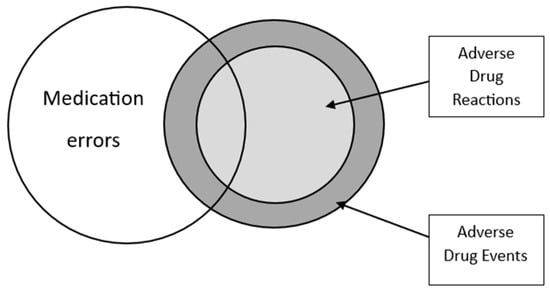
Figure 1
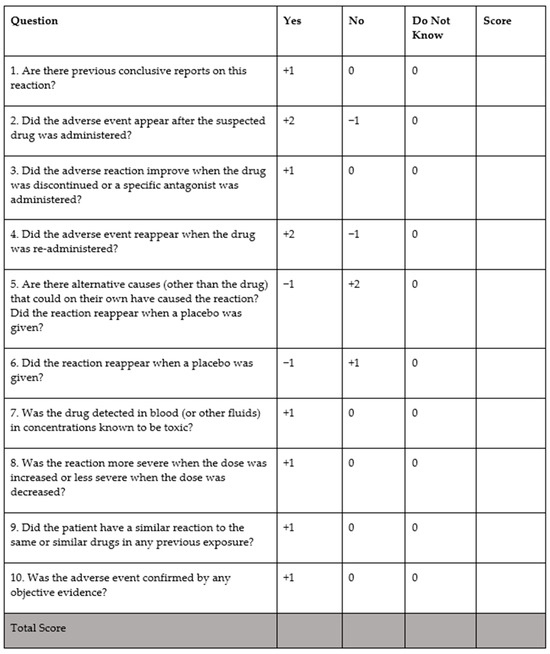
Figure 3
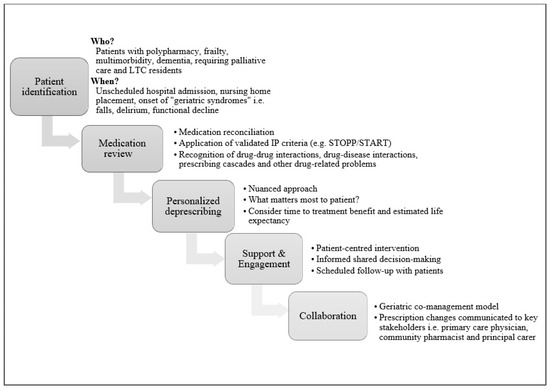
Figure 4
Open AccessArticle
Association of Receipt of Opioid Prescription for Acute Post-Delivery Pain Management with Buprenorphine Discontinuation among Postpartum People with Opioid Use Disorder
by
Taylor N. Hallet, David T. Zhu, Hannah Shadowen, Lillia Thumma, Madison M. Marcus, Amy Salisbury and Caitlin E. Martin
Pharmacoepidemiology 2024, 3(2), 198-207; https://doi.org/10.3390/pharma3020012 - 16 Apr 2024
Abstract
Buprenorphine is a safe and effective medication to treat opioid use disorder (OUD) in pregnant patients and is intended to be continued throughout pregnancy, delivery, and at least the one-year postpartum period. However, delivery often involves the need for acute pain management with
[...] Read more.
Buprenorphine is a safe and effective medication to treat opioid use disorder (OUD) in pregnant patients and is intended to be continued throughout pregnancy, delivery, and at least the one-year postpartum period. However, delivery often involves the need for acute pain management with opioid medications, such as after a cesarean section. For patients receiving buprenorphine, the provision of prescription opioids may negatively impact OUD treatment outcomes; however, not optimally managing acute pain may also impede OUD treatment benefit. Evidence is needed to disentangle the impacts of opioid prescription provision and methods of pain management in the immediate postpartum period on OUD treatment trajectories, ultimately to inform clinical guidelines tailored to the unique needs of pregnant and postpartum people receiving buprenorphine. Accordingly, this study took an initial step towards this goal to conduct a secondary analysis of a retrospective cohort of pregnant patients taking buprenorphine for OUD at the time of delivery (n = 142) to determine whether receipt of an opioid prescription at birth hospitalization discharge was associated with the time of buprenorphine discontinuation within the 12 months following delivery. Among the sample, 26% (n = 37) were prescribed an opioid at the time of birth hospitalization discharge. The number of weeks post-delivery until buprenorphine discontinuation occurred was shorter amongst patients who were prescribed an opioid (median 11 weeks) compared to patients who were not prescribed an opioid (median 39 weeks; p < 0.001 by Mann–Whitney U test). However, a Cox regression model reported that receipt of an opioid prescription following delivery did not significantly increase the hazard ratio for buprenorphine discontinuation. In other words, OUD patients not prescribed an opioid at birth hospitalization discharge continued their buprenorphine for a longer median duration after delivery compared to their counterparts who received prescription opioids; yet, this finding did not reach statistical significance when taking into account additional clinical variables. The findings indicate how further research is warranted to inform evidence-based post-delivery pain practices for postpartum OUD treatment patients.
Full article
(This article belongs to the Special Issue Pharmacoepidemiology and Drug Safety in Pregnancy and Breastfeeding)
►▼
Show Figures

Graphical abstract
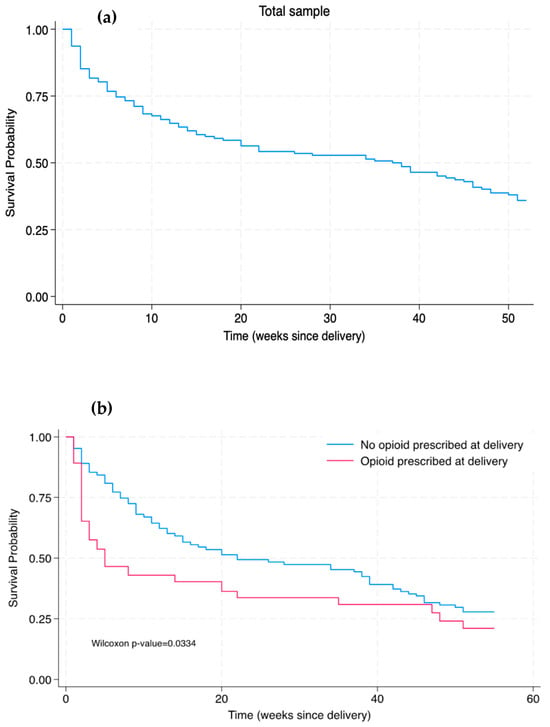
Figure 2
Open AccessArticle
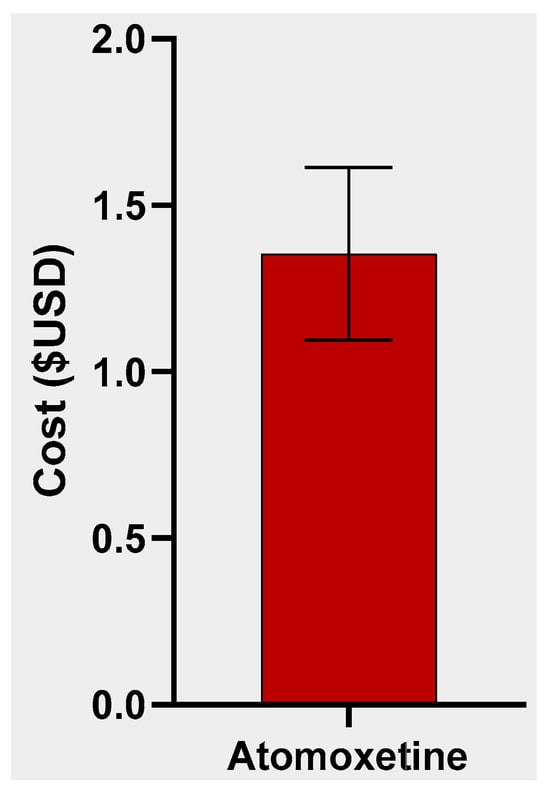
Affordability of Paediatric Oral Anti-Infective Medicines in a Selected District, Sri Lanka
by
Malith Kumarasinghe and Manuj C. Weerasinghe
Pharmacoepidemiology 2024, 3(1), 183-197; https://doi.org/10.3390/pharma3010011 - 12 Mar 2024
Abstract
►▼
Show Figures
In this cross-sectional descriptive study conducted in the Ratnapura district, Sri Lanka, we assessed the affordability of oral pediatric anti-infective medicines (OPAIMs). Using a modified WHO/HAI medicinal price methodology, we examined the availability, median price ratios (MPRs), mean percentage difference, and affordability of
[...] Read more.
In this cross-sectional descriptive study conducted in the Ratnapura district, Sri Lanka, we assessed the affordability of oral pediatric anti-infective medicines (OPAIMs). Using a modified WHO/HAI medicinal price methodology, we examined the availability, median price ratios (MPRs), mean percentage difference, and affordability of the standard treatment of the originator brand (OB) and lowest-priced generic (LPG) OPAIMs in 30 private and 2 state-owned pharmacies. The study revealed disparities in availability, with only 50% of private pharmacies offering all 11 medicinal drugs in their generic form. The MPRs of OPAIMs for OB and LPG varied, with three drugs exceeding the financially acceptable MPR of 2 (albendazole, amoxicillin, and erythromycin). The standard treatment with LPGs costs between 0.17 and 0.85 and between 0.06 and 0.28 days’ wages for the lowest daily salary of the private sector and unskilled public employees, respectively. We identified erythromycin and albendazole as having less than 50% availability in their generic form in private pharmacies. To address these findings, we recommend frequent pricing revisions based on exchange rates and associated costs, coupled with the establishment of a transparent scientific criterion to subsidize essential medicines deemed “unaffordable.” Failure to implement such measures amidst economic crises may adversely impact financial access to essential medications.
Full article
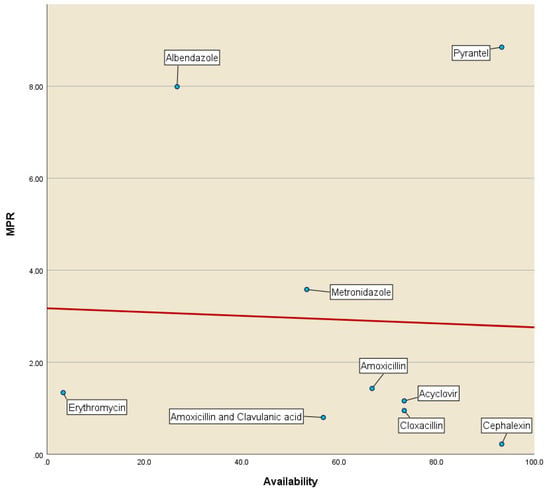
Figure 1
Open AccessArticle
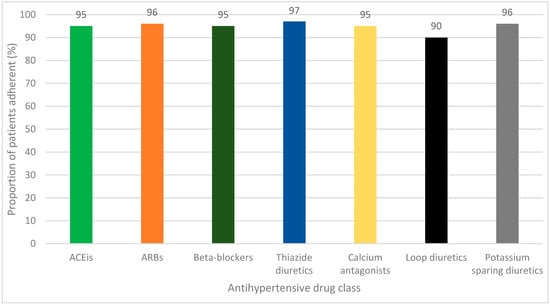
Pioneering Arterial Hypertension Phenotyping on Nationally Aggregated Electronic Health Records
by
Jing Wei Neo, Qihuang Xie, Pei San Ang, Hui Xing Tan, Belinda Foo, Yen Ling Koon, Amelia Ng, Siew Har Tan, Desmond Teo, Mun Yee Tham, Aaron Yap, Nicholas Ng, Celine Wei Ping Loke, Li Fung Peck, Huilin Huang and Sreemanee Raaj Dorajoo
Pharmacoepidemiology 2024, 3(1), 169-182; https://doi.org/10.3390/pharma3010010 - 12 Mar 2024
Abstract
►▼
Show Figures
Background: Hypertension is frequently studied in epidemiological studies that have been conducted using retrospective observational data, either as an outcome or a variable. However, there are few validation studies investigating the accuracy of hypertension phenotyping algorithms in aggregated electronic health record (EHR) data.
[...] Read more.
Background: Hypertension is frequently studied in epidemiological studies that have been conducted using retrospective observational data, either as an outcome or a variable. However, there are few validation studies investigating the accuracy of hypertension phenotyping algorithms in aggregated electronic health record (EHR) data. Methods: Utilizing a centralized repository of inpatient EHR data from Singapore for the period of 2019–2020, a new algorithm that incorporates both diagnostic codes and medication details (Diag+Med) was devised. This algorithm was intended to supplement and improve the diagnostic code-only model (Diag-Only) for the classification of hypertension. We computed various metrics (sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV)) to assess the algorithm’s effectiveness in identifying hypertension on 2813 chart-reviewed records. This pool was composed of two patient cohorts: a random sampling of all inpatient admissions (Random Cohort) and a targeted group with atrial fibrillation diagnoses (AF Cohort). Results: The Diag+Med algorithm was more sensitive at detecting hypertension patients in both cohorts compared to the Diag-Only algorithm (83.8 and 87.6% vs. 68.2 and 66.5% in the Random and AF Cohorts, respectively). These improvements in sensitivity came at minimal costs in terms of PPV reductions (88.2 and 90.3% vs. 91.4 and 94.2%, respectively). Conclusion: The combined use of diagnosis codes and specific antihypertension medication exposure patterns facilitates a more accurate capture of patients with hypertension in a database of aggregated EHRs from diverse healthcare institutions in Singapore. The results presented here allow for the bias correction of risk estimates derived from observational studies involving hypertension.
Full article
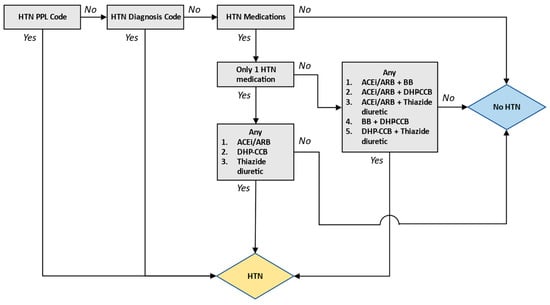
Figure 1
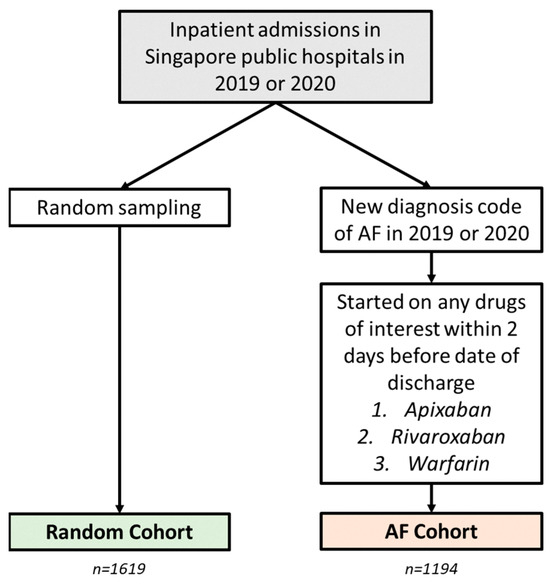
Figure 3
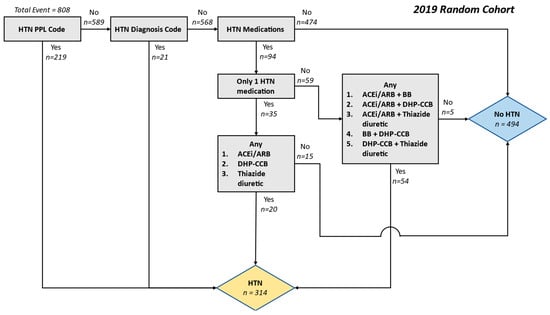
Figure A1
Open AccessReview
Beyond Statins: Novel Lipid-Lowering Agents for Reducing Risk of Atherosclerotic Cardiovascular Disease
by
Teimur Kayani, Bachar Ahmad, Rachel S. Chang, Frank Qian, Melis Sahinoz, Muhammad Waqar Rehan, Antonio Giaimo, Erica S. Spatz and Jiun-Ruey Hu
Pharmacoepidemiology 2024, 3(1), 117-168; https://doi.org/10.3390/pharma3010009 - 5 Mar 2024
Abstract
►▼
Show Figures
Although statins have served as the cornerstone for pharmacological lowering of lipid levels in atherosclerotic cardiovascular disease (ASCVD) risk reduction, many patients are unable to achieve target doses of statin medication due to side effects or target levels of cholesterol reduction on statin
[...] Read more.
Although statins have served as the cornerstone for pharmacological lowering of lipid levels in atherosclerotic cardiovascular disease (ASCVD) risk reduction, many patients are unable to achieve target doses of statin medication due to side effects or target levels of cholesterol reduction on statin monotherapy. The landscape of lipid-lowering strategies has expanded in recent years, with the emergence of therapies that make use of small interfering RNA (siRNA) and antisense oligonucleotides, in addition to traditional small-molecule agents. Non-statin therapies that have shown promising results in randomized controlled trials include adenosine triphosphate-citrate lyase inhibitors, proprotein convertase subtilisin/kexin 9 (PCSK9)-inhibiting antibodies and siRNA, omega-3 polyunsaturated fatty acids, and lipoprotein(a) gene-inhibiting siRNA and ASOs, in addition to older therapies such as ezetimibe. In contrast, cholesteryl ester transfer protein (CETP) inhibitors have shown less promising results in randomized trials. The purpose of this narrative review is to summarize the evidence for these medications, with a focus on phase III randomized trials.
Full article
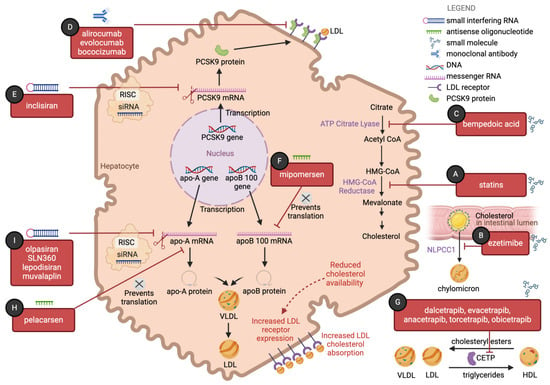
Figure 1